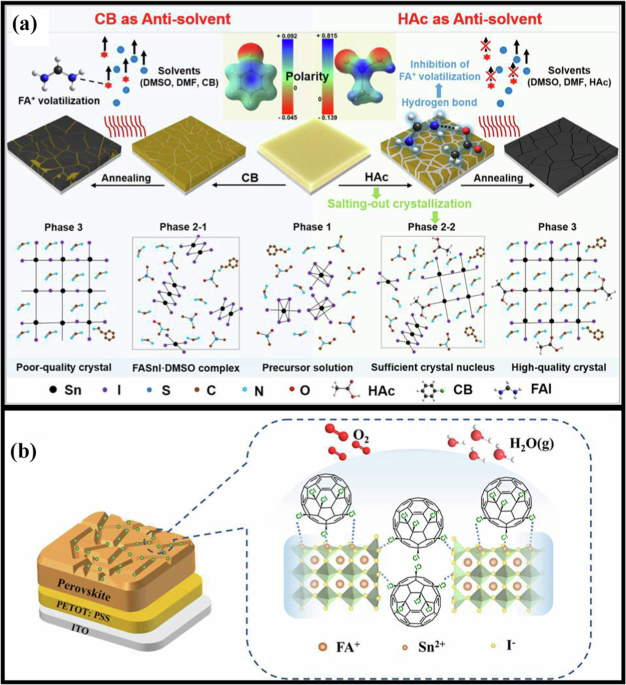
Facet and lattice engineering strategies for PSCs
Designing suitable facet and lattice for perovskites can enhance the performance of PSCs. Utilization of suitable additive can afford the growth of perovskite with suitable facet, and that can enhance the performance of PSCs. Ma et al.75 have demonstrated facet-dependent degradation and facet engineering to achieve stable PSCs. The FAPbI3 with the dominant (100) facet exhibits much higher degradation for moisture when compared to the FAPbI3 with the dominant (111) facet. The key mechanisms of facet-dependent degradation of FAPbI3 films are provided based on theoretical and experimental studies. An elongated Pb−I bond distance is formed due to the strong water adhesion on the FAPbI3 with the dominant (100) facet, and thus a new δ-phase is formed on the (100) facet, which is attributed to the poor stability of the dominant (100) facet. Therefore, a larger surface fraction of the (111) facet is prepared by utilizing cyclohexylamine (boiling point: 134 °C) as an additive to selectively prefer the growth of the (111) facet, and the FAPbI3 with the dominated (111) facet exhibits much higher stability against moisture. The Fig. 2a depicts the XRD pattern of the FAPbI3 with the cyclohexylamine additive, where the FAPbI3 exhibits peaks for both (111) and (100) facets, but the dominant peak is for the (111) facet. A large triangular (111) facet on the individual crystal with fewer lateral (100) facets is observed from the FAPbI3 with cyclohexylamine additive, as shown in Fig. 2b (SEM image), which is contrast to the SEM image of the FAPbI3 with the dominant (100) facet (without additive). The XRD pattern of FAPbI3 with the dominant (111) facet corroborates with the SEM image (the large triangular (111) facet). The exposed-area percentage of the (111) facets from five crystals of the FAPbI3 with and without cyclohexylamine additive is approximately calculated using larger-area SEM images, where the FAPbI3 with cyclohexylamine additive exhibits 85–98% (Fig. 2c) for the (111) facets when compared to that of the control (40–55%). Hence, the FAPbI3 with cyclohexylamine additive exhibits a predominant enlarged (111) facet by diminishing the moisture-sensitive (100) facet, which could be attributed to the selective absorption of the cyclohexylamine additive, and that can diminish the surface energy of the (111) facet. The moisture stability of the FAPbI3 with the cyclohexylamine additive is compared with that of the FAPbI3 without the additive after exposure to 30–40% relative humidity in the air. In the XRD spectra, the FAPbI3 with cyclohexylamine additive exhibits negligible reduction of both (100) peak and dominant (111) peak intensities after exposure for ∼1000 h in moisture (green line; Fig. 2d, top) when compared to that of the unexposed film (black dashed line), which is corroborated with the PL spectra (Fig. 2e, top; a slight peak shift is observed after exposure), where the slight peak shift can be attributed to the phase segregation of FA. Nevertheless, in the XRD spectra, the FAPbI3 without additive exhibits an obvious reduction (~30%) in the dominant (100) peak intensity after exposure for ∼1000 h in moisture (green line; Fig. 2d, bottom) when compared to that of the unexposed film (black dashed line), while a new obvious peak for δ-phase is observed after exposure, which is corroborated with the PL spectra (Fig. 2e, bottom; the obvious reduction in the peak intensity is observed after exposure), which is attributed to the generation of non-radiative recombination centers possibly due to the formation of δ-phase on the (100) facet. Hence, the PSC (FAPbI3 with the dominant (111) facet) exhibits exceptional PCE of 24% (Table 2), with high stability (95% retention for ∼2000 h (Fig. 2f) at 30–40% relative humidity for the device without encapsulation).
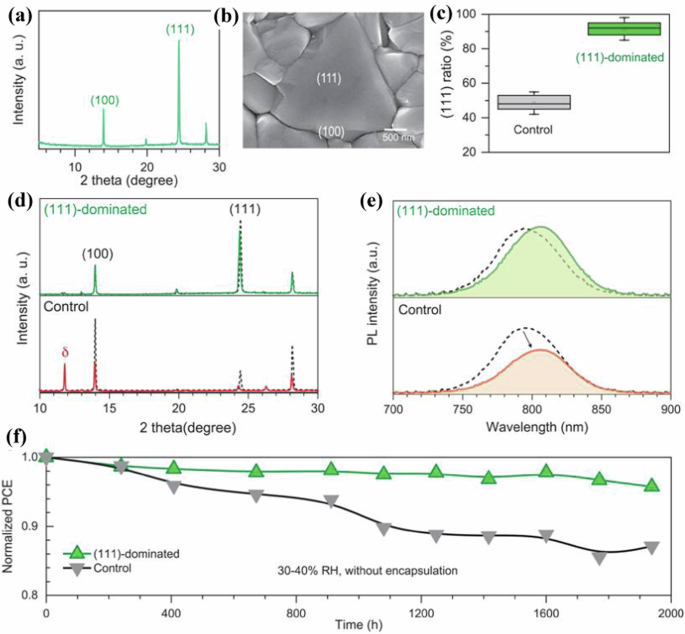
a XRD pattern and b SEM image of the FAPbI3 perovskite thin film synthesized using CHA additive, where the SEM image obviously depicts the large triangular morphology of the (111) facet; c The areal ratio of the (111) facet determined from five crystals of the (111)-dominated perovskite thin films in comparison with the control; d XRD patterns and e PL spectra of the (111)-dominated (top) and control (bottom) samples before and after exposure to humidity for 1000 h (Black dashed line: Before exposure; Red solid lines: Control; Green solid lines: (111)-dominated sample); f Stability test of the unencapsulated devices for the control in comparison with (111)-dominated perovskite stored in air atmosphere under 30–40% RH for 1938 h (reproduced with permission from ref. 75 Copyright 2023, American Association for the Advancement of Science).
The utilization of topochemical assembly strategy can afford the growth of perovskite with suitable orientation, and that can diminish lattice heterogeneity, electronic disorder, and microstructure defects, and that can enhance the performance of the PSCs. Zhu et al.116 have demonstrated that the lattice heterogeneity is minimized by topochemical assembly in polycrystalline halide perovskites to enhance the stability of the PSCs. The mechanisms of topochemical assembly are investigated, where a solid-solid transition is usually observed along the interface of PbI2/perovskite. An alternative pathway for crystal growth along a different coherent interface is achieved due to the introduction of intermediates. Therefore, an optimal (001)-oriented perovskite film is prepared, which possesses reduced lattice heterogeneity, electronic disorder, and microstructure defects. As a result, the PSC exhibits a high PCE of 24.5%, while it exhibits high stability (99% retention for 2500 h by MPP tracking test; 95% retention (one sun, AM 1.5 G) for 500 h (IEC61215 protocols)).
Introduction of pentafluoroanilinium trifluoromethanesulfonate into PbI2 precursor solutions can afford preferred orientation, decrease defects, and enlarge grains, and that can enhance the performance of PSCs. Shao et al.117 have demonstrated the promoted preferential perovskite orientation growth due to the modulation of nucleation and crystallization in PbI2 films by incorporating pentafluoroanilinium trifluoromethanesulfonate into PbI2 precursor solutions to enhance the performance of PSCs. In this case, the modulatory PbI2 films serve as porous templates that promote the formation of perovskites with reduced PbI2 residues, a preferred orientation, decreased defects, and enlarged grains, possibly due to the acceleration of the formation of perovskite and the diminished Gibbs free energy of PbI2. Hence, the PSC exhibits outstanding PCE of 24.52%, while it exhibits high stability (~90% retention for 500 h in an N2 atmosphere at ∼55 °C under constant light irradiation by the MPP tracking test).
Utilization of reconstruction subsurface lattice strategy can afford the growth of perovskite with suitable crystal lattice, modulate the valence band alignment, diminish defects, enhance the carrier-selective transfer and improve the charge carrier lifetime, and that can enhance the performance of PSCs. Gao et al.118 have applied the reconstruction subsurface lattice strategy to achieve stable PSCs; while using this strategy, the positions of the I− and Pb2+ planes are simultaneously modulated. The corner-sharing octahedral is preferred, and high stability is achieved in the crystal lattice by the reconstructed lattice configuration, thereby stabilizing FA-based perovskites. Furthermore, the subsurface lattice reconstruction modulates the valence band alignment and diminishes defects, which enhances the carrier-selective transfer and improves the charge carrier lifetime. Consequently, the PSC exhibits a remarkable PCE of 25.2%, while it exhibits high stability (95% retention for 3000 h under illumination of simulated AM 1.5 by MPP tracking test; 96% retention for 6000 h of storage in a dry box at 25 °C under ∼20% relative humidity).
Various facet/lattice engineering strategies, including utilization of cyclohexylamine as an additive75, topochemical assembly strategy116, incorporating pentafluoroanilinium trifluoromethanesulfonate into PbI2 precursor solutions117, and reconstruction subsurface lattice strategy118, enhanced the efficiency and/or stability of the PSCs.
Strategies for defect/grain engineering and passivation for PSCs
Defect/grain engineering can improve the performance of the PSCs. Tuning the reaction kinetics through anion-π interaction of the AX component of ABX3 perovskite can decrease the defects, and afford high phase purity, and that can enhance the performance of the PSCs. Huang et al.67 have tuned the reaction kinetics through anion-π interaction of the AX component of ABX3 perovskite with hexafluorobenzene, which is an alternative strategy to conventional methods that rely on the BX2 component. These two strategies are independent, but they work together to attain ‘dual-site regulation’, which leads to delicate control over the reaction of AX with BX2 without unwanted intermediates. As a result, the FAPbI3 perovskite film affords red-shifted absorption, lesser defects, and high phase purity without measurable nanoscale δ phase. Hence, the PSC exhibits a superior PCE of 26.07% and excellent stability (94% retention for 1258 h by MPP tracking test).
Lattice strains and phase transitions can be induced in halide perovskites by the variations in the daily temperature, and that can affect the stability of the PSCs. Therefore, developing PSCs that can be stable at variable temperature (ambient, low, and high temperature) is highly desirable. The introduction of suitable polymer with ordered dipolar structure into the perovskite precursor can stabilize the perovskite black phase, promote the growth of a low-defect crystalline film, generate the dipoles at the surface of the perovskite, diminish ion migration, enhance interfacial charge extraction, enhance hydrophobicity, and that can enhance the performance of PSCs at variable temperature. Li et al.78 have utilized the ordered dipolar structure of β-poly(1,1-difluoroethylene) (β-pV2F) for the stabilization of the perovskite black phase to enhance the performance of PSC by controlling the energy alignment and perovskite film crystallization. Therefore, the PSC exhibits enhanced PCE of 24.6% (Table 3), and it exhibits high stability (88 and 96% retention for 1000 h by MPP tracking test at 75° and 25 °C, respectively), while negligible signs of fatigue are observed for the PSCs at rapid thermal cycling between +80 °C and −60 °C, suggesting its high thermal cycling stability. The β-pV2F exhibits an ordered molecular dipole distribution due to the polymeric backbone of the β-pV2F and alternate symmetric hydro and fluorocarbon units. β-pV2F with 0.5 mg/mL (molecular weight: 180,000) had been used. Figure 3 depicts the role of β-pV2F on the work function and film morphology. The control perovskite film exhibits evident voids at the grain boundaries (average grain size: ~400 nm; Fig. 3a−c), where non-radiative recombination centers and shunting paths can be formed due to these defects. In contrast, the β-pV2F-treated perovskite film exhibits high density with large grain size (~480 nm; Fig. 3d−f). The XRD of the β-pV2F-treated perovskite film exhibits improved crystallinity as inferred from a smaller full width at half maximum in the (001) peak. The atomic force microscopy studies disclose that the β-pV2F-treated perovskite film exhibits a lesser surface roughness of 41.1 nm when compared to that of the control (54.4 nm), which can ameliorate coverage with charge-transporting layers. The adjacent hydrogen atoms in β-pV2F can have a partial positive charge density due to the fluorine atoms with electron-withdrawing effect. Furthermore, the β-pV2F exhibits an all-trans planar zigzag conformation, which is almost similar to a Lewis acid, and that can interact with the surface of the perovskite. In the FTIR spectra, the β-pV2F-treated perovskite film exhibits the –CH2 stretching vibration peak at 3019 cm−1 when compared to that of bare β-pV2F (3025 cm−1), possibly due to a solid C-H···X dipole interaction of –CH2 moieties with halide ions of the [PbX6]4− frame. Thus, the β-pV2F-treated perovskite film exhibits an upward shift of the surface work function (Fig. 3g), possibly due to the change in crystallization during film growth caused by the aforementioned polar interaction between β-pV2F and precursors. Therefore, the β-pV2F-treated perovskite film exhibits an increase in surface work function up to 300 meV (Fig. 3h) when compared to that of the control, and that can enhance the interfacial charge extraction and improve the stability of PSC.
Schematic diagram illustrates the processing of a control and d target perovskites; SEM images of b top view and c cross section of control perovskites; SEM images of e top view and f cross section of target perovskites; g, h work function (WF) shift associated with the perovskite functionalized by β-pV2F (reproduced with permission from ref. 78 Copyright 2023, American Association for the Advancement of Science).
Effective rejuvenated of aged perovskite quantum dots can be achieved through in situ defect healing treatment. Chen et al.119 have effectively rejuvenated aged perovskite quantum dots (PQDs) using in situ defect healing treatment (DHT). Theoretical calculations and experimental studies disclose that the surface matrix defects of aged PQDs could be strongly anchored by the I3− anions generated from tetra-n-octylammonium iodide and iodine, which can considerably reduce the non-radiative recombination of photogenerated charge carriers. Furthermore, the morphology of aged PQDs could be renovated by DHT, which can enhance the stacking orientation of PQD solids and considerably ameliorate charge carrier transport within PQD solids. As a result, the PSC exhibits a high PCE of 15.88%, while it exhibits high stability (60% retention for 200 h by MPP tracking test).
Utilization of suitable grain modulator can consolidate the crystal structure, while utilization of redox-active radical polymer can effectively p-dope the hole-transporting material through hole injection, and that can enhance the performance of the PSCs. You et al.68 have performed two strategies (grain modulation and radical polymeric p-doping) to enhance the performance of the PSC. A phosphonic acid-functionalized fullerene derivative is introduced in mixed-cation perovskites as a modulator for the grain boundary to consolidate the crystal structure, thereby improving the resistance of the perovskite sheet against heat, moisture, and illumination. Furthermore, the poly(oxoammonium salt) as a redox-active radical polymer is used to effectively p-dope the hole-transporting material through hole injection, which also inhibits the diffusion of lithium ions. Hence, the PSC and mini-module demonstrate satisfactory PCE of 23.5% (1 cm2) and 21.4% (17.1 cm2), respectively, while maintains high stability (95.5% retention for 3265 h by MPP tracking test at 70° ± 5 °C).
Several strategies have been applied to enhance the passivation of the perovskites to enhance the performance of PSCs. The library of low-dimensional capping materials with metal cations can be expanded by processing a full precursor solution with both metal and ammonium halides rather than the commonly used Pb2+ and Sn2+, where a zero-dimensional zinc-based halogenometallate can induce stronger n–N isotype three-dimensional/low-dimensional heterojunctions and higher surface passivation when compared to its lead-based counterpart, and that can enhance the performance of the PSCs. Ye et al.69 have expanded the library of low-dimensional capping materials with metal cations rather than the commonly used Pb2+ and Sn2+ by processing a full precursor solution with both metal and ammonium halides. Thus, facile synthetic control is achieved for the low-dimensional capping layer, while greater versatility is attained for low-dimensional interface engineering. Therefore, a 0D PEA2ZnX4 (zinc-based halogenometallate; PEA = phenethylammonium, X = Cl/I) exhibits much higher surface passivation and robust n-N isotype 3D/low-dimensional heterojunctions when compared to that of lead-based counterpart. The resulted PSC exhibits a superior PCE of 24.1% with high stability (94.5% retention for 1009 h by MPP tracking test).
Utilization of dimethylacridine-based molecular doping process can effectively passivate the grain boundaries, and that can enhance the performance of the PSCs. Tan et al.120 have demonstrated a dimethylacridine-based molecular doping process to build a well-matched p-perovskite/ITO contact with passivation of grain boundaries to attain enhanced performance for PSCs. Thus, a molecule-extrusion process occurs, where the molecules can be extruded to the grain boundaries from the precursor solution, while the crystallization process occurs at the bottom of the film surface in the chlorobenzene-quenching process. The core coordination complex of lead polyiodide of perovskite with the deprotonated phosphonic acid group of the molecule is accountable for both electronic charge transfer and mechanical absorption, and that cause p-type doping of the perovskite film. As a result, the PSC exhibits a high PCE of 25.86%, while it exhibits high stability (96.6% retention for 1000 h by MPP tracking test).
Utilization of pseudo-halide anion engineering strategy can effectively passivate the surface, and that can enhance the performance of the PSCs. Xu et al.121 have performed pseudo-halide anion engineering for bifunctional surface passivation to enhance the performance of PSCs, while a machine learning workflow is demonstrated to facilitate the discovery process through full-DFT calculations for training the model. Thus, promising molecules with a head group can be identified through the physics-informed machine learning model, and that inhibits anti-site defect generation and lattice distortion, while a tail group is optimized for robust attachment to the surface. Furthermore, 15 potential bifunctional pseudo-halide anions having the ability to passivate both acceptors and donors are identified, where the most effective passivant is sodium thioglycolate. As a consequence, this strategy contributes to PSC with unexceptional PCE of 24.56% and robust stability (96% retention for 900 h by MPP tracking test).
Utilization of ligand reactivity engineering strategy can afford stable PSCs at high-temperatures. Park et al.122 have demonstrated ligand reactivity engineering to achieve stable PSCs at high-temperatures. Ammonium ligands are utilized, which are non-reactive with the bulk of perovskites, while a library is determined that alters ligand molecular structure systematically. Thus, fluorinated aniliniums exhibit interfacial passivation and, at the same-time, reduce reactivity with perovskites. Hence, the PSC exhibits a large PCE of 24.09%, while it exhibits high stability (85% retention for 1560 h by MPP tracking test at 85 °C and 50% relative humidity).
Introduction of insulator layer having optimal thickness with random nanoscale openings between the absorber and transport layer can diminish surface recombination, while the crystallinity and bulk recombination lifetime can also be enhanced, and that can enhance the performance of the PSCs. Peng et al.123 have introduced ∼100-nm-thick insulator layers with random nanoscale openings between the absorber and transport layer without deteriorating both the open-circuit voltage (Voc) and the fill factor (FF) to diminish surface recombination and achieve high-performance PSCs. Drift-diffusion simulations are conducted for cells having this porous-insulator contact and comprehended with a solution process through managing the growth mode of Al2O3 nanoplates. Thus, the 87.9% of the Shockley-Queisser limit is the product of Voc × FF, while the surface recombination velocity was diminished to 9.2 cm s−1 from 64.2 cm s−1 at the p-type contact, and the bulk recombination lifetime is elevated to 6.0 μs from 1.2 μs due to the enhancement in the crytallinity of perovskite. As a result, the PSC with a porous-insulator contact with a ∼25% diminished contact area exhibits a high PCE of 25.56%, while it exhibits high stability (>98% retention for 1000 h by MPP tracking test).
Utilization of in situ tin selenide deposition strategy can enhance the surface crystallization and diminish the number of surface traps, and that can enhance the performance of PSCs. Chen et al.124 have obtained PEA2(FA0.5MA0.5)4(Pb0.5Sn0.5)5I16 (n = 5) with a gradient structure through a two-step blade coating. Furthermore, improved surface crystallization and a diminished number of surface traps are observed for the perovskite films after treatment with tin (II) acetate and N,N-dimethylselenourea, due to the in situ generation of tin selenide, where the active layer is well protected from the environment by this hydrophobic tin selenide. Therefore, the PSC exhibits PCE of 15.06% with a VOC of 0.855 V and negligible hysteresis, while it exhibits high stability (100% retention for 15 h by MPP tracking test).
Utilization of crystallization strategy can passivate the defects and enhance the crystallization, and that can enhance the performance of the PSCs. Zhang et al.125 have demonstrated holistic defect passivation and crystallization manipulation through the crystallization strategy via a novel intermediate phase to construct high-quality perovskite films to achieve high-performance PSCs. Thus, the orientational growth of the α-phase perovskite is promoted during crystallization after introducing phenformin hydrochloride (PFCl) into the precursor solution, where the intermediate phase PFCl•FAI diminishes the formation of the δ yellow phase. Furthermore, a post-treatment is performed using multiactive-site S-methylisothiosemicarbazide hydroiodide, leading to a reduction in the bulk and interfacial trap-assisted non-radiative recombination losses, which is attributed to diminished defects, enhanced crystallization, and released residual stress. As a result, the PSC exhibits a high PCE of 24.67%, while it exhibits high light and thermal stability (94% retention for 600 h by MPP tracking test; 84% retention for 1008 h at constant light illumination; 90% retention for 864 h at constant heating at 85 °C).
Utilization of surface n-type band bending strategy can enable the p to n-type transition at the surface and passivate the defects, and that can enhance the performance of the PSCs. Wang et al.126 have demonstrated a surface n-type band bending strategy to alleviate the severe energy losses in inverted PSCs at the interface between the n-type ETL and p-type perovskite. Furthermore, a p to n-type transition is observed at the surface of the CsPbI3 due to the post-deposition of propylamine hydrochloride to expedite the extraction and separation of the electrons while passivating the defects of the surface. Hence, the PSC exhibits an outstanding PCE of 20.17% (38.93% at indoor light illumination using LED with 307 mW cm−2 having 1000 lux) with robust stability (∼97% retention for 792 h in light soaking at the open circuit condition).
The α-phase of FAPbI3 can be stabilized without band gap broadening by utilizing suitable cations and anions. Yoo et al.127 have utilized R4N+ cations and Cl− anions for the stabilization of the α-phase of FAPbI3 without band gap broadening. The resulted PSC and module exhibit unexpected PCE of 24.53% and 21.23% (36.4 cm2), respectively, while the perovskite solar module exhibits high stability (93% retention for 870 h at constant one-sun illumination).
The utilization of two alkylammonium halide modulators can inhibit halide phase segregation and enhance passivation, and that can enhance the performance of the PSCs. In addition, Alharbi et al.128 have utilized two alkylammonium halide modulators to enhance the stability and PCE of PSCs based on FA0.65MA0.35Pb(I0.65Br0.35)3 and α-FAPbI3, where the modulators inhibit halide phase segregation induced by light in the former. The 2D NMR studies disclose that the modulators are found at grain boundaries and surfaces, while the computational studies reveal that the modulators exhibit a higher passivation effect when compared to that of established compounds. Therefore, the PSC based on α-FAPbI3 exhibits a high PCE of 24.9%, while it exhibits high stability (∼90% retention for 1200 h by MPP tracking test).
The utilization of suitable surface passivation can enhance the performance of the PSCs. Hieulle et al.129 have discovered the effect of white light in the degradation of the surface of FAPbI3-rich perovskite absorbers through computational studies and multiscale characterization techniques. Thus, the degradation kinetics are investigated, where the chemical degradation of perovskite into secondary phases with metallic I-V characteristics and a larger work function is triggered by the white light. In addition, the degradation of perovskite has been triggered by a synergetic effect of the defects in perovskite and the illumination of light. In addition, surface passivation is performed to determine the effect of defect sensitivity in the degradation process. Furthermore, the light-induced degradation of perovskite with respect to the wavelength is determined by utilizing filtered light. Therefore, the PSC exhibits a large PCE of 22%, while it exhibits high stability (75% retention for 500 h by MPP tracking test).
The utilization of solid-phase passivation strategy can alleviate the usage of solvents for passivation, enhance the passivation and diminish the carrier non-radiative recombination loss, and that can enhance the performance of the PSCs. Luo et al.130 have utilized 2-mercaptopyridine as a passivator on pre-prepared solid-state films to bond surface uncoordinated Pb2+ defects through a solvent-free solid-phase passivation (SPP) route rather than the conventional solution processing route. Furthermore, owing to the robust coordination ability of the passivator with the uncoordinated Pb2+ defects, the passivator is found to anchor on the defect sites of the surface of the perovskite film even if the passivator film is destroyed. Thus, the defect passivation considerably diminishes the carrier non-radiative recombination loss. Hence, the PSC exhibits an excellent PCE of 25.2%, a high VOC of 1.20 V, and a low voltage loss of 0.34 V, while it exhibits high stability (92% retention for 800 h by MPP tracking test).
Utilization of organic iodides can enhance the energy barrier for iodine interstitial migration and improve the defect passivation, and that can enhance the performance of the PSCs. Ren et al.131 have utilized organic iodides to enhance the performance of the PSCs. Thus, the energy barrier for iodine interstitial (Ii) migration is increased by the organic iodides with suitable cations, possibly due to their strong interaction with the surface of the perovskite, where the interaction emerges from a high dipole moment and a stable steric alignment. The role of four classical phenylalkylammonium iodides on Ii migration is studied, where the length of the alkyl chain of cations possesses influence on the inhibition of Ii migration. As a result of Ii migration inhibition and defect passivation on the surface of the perovskite film, the phenylpropylammonium iodide-treated PSC exhibits a high PCE of 24.2%, while it exhibits high stability (92% retention for 240 h by MPP tracking test).
Utilization of phase transition engineering strategy can improve the crystallinity, and modulate the energy levels, conductivity, carrier mobility, and trap states, and that can enhance the performance of the PSCs. Chen et al.132 have utilized a holistic strategy to enhance carrier transportation in a SnO2/perovskite stack. At first, the NT:SnO2 is obtained by co-doping Nb5+ and Ta5+ with SnO2, which can modulate the energy levels, conductivity, carrier mobility, and trap states, while the NT:SnO2 tunes the crystallization process of the perovskites, leading to improved crystallinity. Furthermore, carrier lifetime is increased to >3 μs, and non-radiative recombination is considerably decreased after the introduction of 4,4’-dithiodibutyric acid as a multifunctional passivator molecule to subsequently passivate the charged traps in perovskites. As a result, the PSC exhibits satisfactory PCE of 25.3%, while it exhibits high stability (90% retention for 1200 h by MPP tracking test).
Utilization of interfacial-engineering method can effectively passivate the defects and diminish the trap densities, and that can enhance the performance of the PSCs. Kim et al.133 have demonstrated an interfacial-engineering approach containing the in situ thermal phase transition of AAFos (AAFos: alkylammonium formates) to effectively passivate the defects to enhance the performance of PSCs. AAFos is composed of a big organic cation with a pseudohalide anion, which can be involved at relatively low temperatures in the passivation process as the liquid phase due to the weak coordination between the anions and cations. Thus, the AAFos exhibits several merits in interfacial engineering: (a) The grain boundaries of perovskites can be effectively passivated by the AAFos to diminish the trap densities by improving the liquid-like behavior of the AAFo. (b) The effective passivation at iodide vacancies is achieved to enhance the thermal stability due to the formate anion, where the fromate anion has a comparatively larger affinity with iodide vacancies when compared to that of other halides. (c) The moisture stability is enhanced by the decylammonium cations with lengthy alkyl chain which inhibits the permeation of moisture into the perovskite layer. Therefore, the PSC and module exhibit high PCE of 25% and 20.82% (23.75 cm2), respectively, while the PSC exhibits high thermal and moisture stability.
Utilization of suitable aging-resistant precursor can afford higher crystallinity and larger grain size, and that can enhance the performance of the PSCs. Huang et al.134 have added 2,3,5-trichlorobenzaldehyde with chloride ions and a C═O group (3Cl-BZH; Fig. 4a) into the solution (organic salt precursor) to alleviate the excess organic amine formation by Schiff-base reactions, and that enhances the stability of the precursor solution and prevents further irreversible amine-cation reactions of formamidine-methylammonium (FA-MA+). The FTIR is performed to investigate the interaction of 3Cl-BZH with organic salt (Fig. 4b, c). The stretching vibration peak of C═O exhibits a shift to 1638 cm−1 from 1628 cm−1, and the stretching vibration peak of N─H exhibits a shift to 3451 cm−1 from 3438 cm−1 after mixing FAI with 3Cl-BZH, suggesting the existence of a chemical interaction of the C═O group of 3Cl-BZH with FA+. Thus, slower black phase formation is occurring, possibly due to the delay of the reaction of FAI with PbI2 after the addition of 3Cl-BZH. Therefore, a higher crystallinity and larger grain size of perovskite are obtained due to the delayed organic ion release and slow reaction when compared to the original unannealed film, as shown in Fig. 4d. Figure 4e depicts the schematic diagram that illustrates the role of 3Cl-BZH on the crystallization process of perovskite films. If 3Cl-BZH is not used, a rapid reaction can occur between FAI and PbI2, which can form a perovskite layer on top of PbI2, inhibit the diffusion of FAI into the inner PbI2 layer, and provide a perovskite phase with low crystallinity and excess PbI2 residues. In contrast, the diffusion rate of FAI within the PbI2 matrix has been slowed down after the introduction of 3Cl-BZH, leading to the gradual crystallization of perovskite nuclei, while the interaction of 3Cl-BZH with FAI enhances the perovskite reaction, resulting in the generation of an orderly and uniform organic salt layer, leading to the formation of high-quality perovskite films. Therefore, the PSC with 3Cl-BZH (with defect passivation effect) using a conventional annealing process exhibits a high PCE of 24.08% and high stability, while it exhibits a high PCE of 22.01% after broadening the annealing window of the wet perovskite sheet in the air atmosphere to 240 h, suggesting its prospects towards commercialization.
a Chemical structure of 2,3,5-trichlorobenzaldehyde (3Cl-BZH); b FTIR spectrum of FAI mixed with 3Cl-BZH solution in comparison with 3Cl-BZH solution; c FTIR spectrum of FAI mixed with 3Cl-BZH solution in comparison with FAI solution; d Schematic diagram illustrates the synthesis process of perovskite films with and without 3Cl-BZH; e Schematic diagram illustrates the crystallization of perovskite films with and without 3Cl-BZH (reproduced with permission from ref. 134 Copyright 2023, Wiley-VCH GmbH).
Utilization of room-temperature molten salt-mediated perovskite growth strategy using dimethylamine formate can afford low-defect density and a large grain size, and that can enhance the performance of the PSCs. Zai et al.135 have added dimethylamine formate (DMAFa, a room-temperature molten salt) into the precursor solution to control the crystallization behavior of CsPbI3 perovskite films. The coordination of DMAFa with Pb2+ can form HCOO−-Pb2+ in the initial stage, and subsequent annealing gradually decompose the HCOO−-Pb2+ into I−-Pb2+, leading to a delay in crystallization rate, while the defects of the perovskite films can be passivated by the interaction of DMA+ with the uncoordinated Pb2+, and that generates a high-quality CsPbI3 film with a low-defect density and a large grain size. As a result, the PSC exhibits a high PCE of 20.4% while it exhibits high stability.
In situ polymerization of cross-linked perovskite-polymer composites can enhance the crystallinity and increase the grain size, and that can enhance the performance of the PSCs. Guo et al.136 have demonstrated cross-linked perovskite-polymer composites with in situ polymerization to enhance the performance of PSCs, where the acrylamide monomers are cross-linked with the assistance of light to the non-crystalline perovskite films. The preferred crystal orientation in the polycrystalline perovskite films is induced by the acrylamide monomers, and the perovskite grain size is enlarged while the perovskite grains are cross-linked. The cross-linked interfacial layer protects the perovskite film against water corrosion, where this layer acts as an airtight wall. Furthermore, the lattice strain during perovskite-film crystallization is effectively released by the liquid properties of acrylamide. Hence, the PSC and module exhibit remarkable PCE of 24.45% and 20.31% (33 cm2), respectively, and the PSC exhibits high stability, while it exhibits a high VOC of 1.199 V, which is a higher VOC than that previously reported for promising hybrid PSCs with TiO2 as ETLs.
The utilization of suitable ionic liquid as co-solvent can modulate the crystallinity, and afford interfacial band alignment and effective charge transport of the screen-printed film, and that can enhance the performance of the PSCs. Chen et al.137 have demonstrated the fabrication of fully screen-printed perovskite solar cells with a high PCE of 16.95% towards commercialization through modulating confined perovskite crystallization within the mesoporous layer. The complete volatilization of ionic liquid methylamine acetate (MAAc) is achieved, and perovskite crystals with high filling degree within the mesoporous structure are attained due to the addition of ionic liquid MAPa (MAPa: methylamine propionate) with robust coordination as a co-solvent to stimulate the escape of MAAc molecules, where volatilization channels for solvent are generated in a confined mesoporous structure. Additionally, interfacial band alignment and effective charge transport of the screen-printed film are achieved due to the MAPa, where the vertical growth of perovskite crystals is stimulated by MAPa and the unbonded Pb2+ is coordinated by MAPa on the surface of the perovskite. Hence, the PSC exhibits a high PCE of 16.95%, while it exhibits high stability (∼85.3% retention for 250 h by MPP tracking test).
Utilization of halogenated phenylethylammonium iodide salts can effectively tune the passivation of the perovskite, while the meta-substituted cations can afford better performance than that of ortho and para counterparts, which could be due to the higher interfacial dipoles and lower formation energies, and that could enhance the performance of the PSCs. Gozukara Karabag et al.138 have modulated the passivation of 2D perovskite, while the role of steric and electronic effects on the performance of 3D/2D PSCs has been investigated. Thus, several halogenated PEA+ iodide salts are prepared through the facile route, and the salts are deposited on the top of 3D perovskite, where the salts are x-XPEAI (x is ortho (o), meta (m), or para (p); X is F, Cl, or Br). The grazing-incidence wide-angle X-ray scattering analyses and XRD investigation disclose the formation of a 2D perovskite layer for all cations, irrespective of the position and nature of the halogen. Theoretical calculations disclose that m-substituted cations exhibit higher performance when compared to their o– and p– counterparts owing to their higher interfacial dipoles and lower formation energies. Therefore, the PSC with m-BrPEAI exhibits a high PCE of 23.42%, while the PSC with m-ClPEAI exhibits high stability (87% retention for 1000 h by MPP tracking test).
The utilization of β-alaninamide hydrochloride can generate in situ formation of 2D perovskite seeding, which can enhance the quality of crystallization, provide uniformity of perovskite film, and that can enhance the performance of indoor PSCs. Li et al.139 have utilized β-alaninamide hydrochloride, which spontaneously generates a layer of 2D perovskite nucleation seeds, to enhance the quality of crystallization, uniformity of film, and performance of indoor PSCs. The charge carrier lifetime is increased 10-fold after the introduction of β-alaninamide hydrochloride due to the enhanced film quality with lesser lattice stress, smaller trap density, and better uniformity. Hence, the indoor PSCs presents impressive PCE of 42.12% (0.09 cm2), 40.93% (1.0 cm2), and 40.07% (2.56 cm2) with light intensity of 1000.52, 1000.69, and 1000.85 lux, respectively, with LED illumination, while it exhibits high stability.
Utilization of uracil as a binder in the perovskite film can strengthen grain boundaries, effectively passivate defects, strengthen the interface between ETL and perovskite, and that can enhance the performance of the PSCs. Wang et al.79 have utilized uracil as a binder in the perovskite film to improve the performance of PSCs. The uracil can strengthen grain boundaries and effectively passivate defects to improve the stability of perovskite films, while it strengthens the interface between ETL (SnO2) and perovskite to enhance the binding force. Hence, the PSC exhibits a high PCE of 24.23% with negligible hysteresis and high reproducibility, while it exhibits robust stability (90% retention for 5960 h by MPP tracking test) and high reproducibility.
Utilization of surface in situ reconstruction strategy using CsF treatment can enhance hole extraction, inhibit non-radiative recombination, effectively passivate the surface defects, and that can enhance the performance of the PSCs. Chu et al.140 have demonstrated a surface in situ reconstruction strategy to enhance the efficiency of the PSC through CsF treatment for inorganic perovskite to enhance hole extraction and inhibit non-radiative recombination, while the mechanism has been verified by experiments and kinetic calculations. Thus, effective passivation of surface defects can be achieved after the incorporation of fluorine, while a prolonged carrier lifetime of 737.2 ns is obtained from 11.5 ns. Additionally, a wider-bandgap perovskite layer can be formed as a graded heterojunction to enhance hole extraction. As a result, the PSC exhibits a high PCE of 21.02%, a fill factor of 85.3%, and an open-circuit voltage of 1.27 V, while it exhibits high stability (86% retention for 400 h by MPP tracking test).
Strategies for defect/grain engineering67,68,78,119 and passivation129 including expansion of the library of low-dimensional capping materials with metal cations69, dimethylacridine-based molecular doping process120, pseudo-halide anion engineering121, introduction of insulator layer having optimal thickness with random nanoscale openings between the absorber and transport layer123, in situ tin selenide deposition124, crystallization strategy125, surface n-type band bending strategy126, utilization of R4N+ cations and Cl− anions127, utilization of two alkylammonium halide modulators128, solvent-free solid-phase passivation route130, utilization of organic iodides131, phase transition engineering132, interfacial-engineering method133, utilization of aging-resistant precursor134, room-temperature molten salt-mediated perovskite growth135, in situ polymerization of cross-linked perovskite-polymer composites136, modulation of confined perovskite crystallization137, modulation of the passivation of 2D perovskite138, in situ formation of 2D perovskite seeding139, utilization of uracil as a binder into the perovskite film79, and surface in situ reconstruction strategy140 enhanced the efficiency and/or stability of the PSCs.
Dion–Jacobson/hybrid perovskite for PSCs
Designing suitable Dion–Jacobson (DJ)/hybrid perovskite can enhance the performance of PSCs. Utilization of triple-solvent engineering strategy can afford pure-phase, finely oriented crystal grains and a homogenous quantum well width structure for DJ-2D perovskites, and that can enhance the performance of the PSCs. Ahmad et al.74 have designed 4APP(FA)3Pb4I13-based quasi-DJ-2D perovskite (4APP: 4-aminopiperidinium) to improve the stability of the PSCs. The 4APP possesses strong rigidity and robust hydrogen bonding ability with the perovskite, leading to an obvious increase in the intrinsic stability of the perovskite. The DJ-2D perovskites with pure-phase, finely oriented crystal grains and a homogenous quantum well width structure are obtained through the triple-solvent engineering strategy by coordinating solvent properties. Thus, the PCE of the unencapsulated PSCs is improved by 48% using MA-free DJ 2D perovskite (n ≤ 4). As a result, the PSC exhibits a high PCE of 17.27% (Table 4), while it exhibits high stability (95% retention for 2000 h at 40 °C by MPP tracking test for the unencapsulated device; 95% retention for >3000 h in air at 85 °C with 60–90% relative humidity).
The performance of the PSCs can be improved using 2D/3D heterostructure, where the amidino based DJ 2D perovskite can afford skeleton for 3D growth upon the dissolution-recrystallization process, which can diminish bulk defects and alleviate volume collapse. Yang et al.141 have utilized amidino-based Dion–Jacobson 2D perovskite to achieve stable and efficient FAPbI3-based 2D/3D heterostructure in PSCs. Time-resolved optical diagnostics is utilized to investigate the effect of DJ 2D perovskite on the nucleation and growth mechanisms and crystallization pathways of 3D perovskite. The crystallization process of the 2D/3D perovskite in comparison with the neat 3D perovskite is illustrated in Fig. 5. FA-rich α-phase perovskites are formed by the preferential crystallization of 3D perovskite during the following perovskite film fabrication processes, including spin coating, dissolution, and recrystallization, when the entrapped DMSO eliminates the bulk film in the course of thermal annealing. Thus, void generation at grain boundaries on 3D perovskite induced by the volume collapse of the crystals is observed due to the evaporation of DMSO and the uniform nucleation in the course of dissolution-recrystallization equilibrium. In contrast, the nucleation and growth pathways are altered and the volume collapse is inhibited by the DJ 2D perovskite, where a high-quality perovskite film with suppressed bulk defects and voids is obtained due to the modulated crystallization process, leading to efficient charge transport. Hence, the PSCs exhibit a high PCE of 24.9% (on rigid substrate) and 22.3% (on flexible substrate), while the PSC exhibits high stability, and high bending cycle stability for flexible solar cells (92% retention for 3000 bending cycles).
A schematic diagram illustrates the mechanism of crystallization of perovskite films (reproduced with permission from ref. 141 Copyright 2023, Elsevier Inc.).
Utilization of suitable solvent and ligand for quasi-2D-DJ phases on 3D perovskites can enhance the performance of the PSCs. Ramakrishnan et al.142 have utilized 2,2,2-trifluoroethanol as a solvent and 3-aminomethylpyridine as a ligand for the fabrication of quasi-2D DJ phases on 3D perovskites to achieve enhanced performance for PSCs. In situ X-ray scattering is used to determine the structural evolution and solvent-mediated formation dynamics of 2D and 3D heterostructures. The spatial distribution of quasi-2D phases for n is from 2 to 4, and the formation sequence is determined by the interaction of the solvent with the 3D surface. When isoproponol is used as a solvent, the perovskite is reconstructed into a PbI2-rich surface, where a strata is generated with a smaller n first, and then a thinner substratum is formed with a larger n. However, when 2,2,2-trifluoroethanol is used as a solvent, the 3D surface is protected, thereby enhancing the generation of homogenously distributed larger n domains first and smaller n last. Thus, DJ perovskites have been utilized with excellent charge transport properties and structural robustness to construct 2D/3D heterostructures with n ≥ 3 and design a suitable energy landscape for electron tunneling. Hence, the PSC exhibits impressive PCE of 23.6%, with high stability (94% retention for 960 h at 85 °C).
Fabrication of organic-inorganic hybrid perovskite by incorporating a tiny amount of organic cations for inorganic cations can increase the optoelectronic properties, and that can enhance the performance of the PSCs. Jiang et al.143 have prepared tetragonal (β-) (DMA, Cs)PbI3 by incorporating a tiny amount of DMA+ (large organic cation) for Cs+ with a small ionic radius, while the obvious evidence for the generation of organic-inorganic hybrids is provided. The hybrid organic-inorganic β-(DMA, Cs)PbI3 with a dominant inorganic composition exhibits higher optoelectronic properties when compared to that of inorganic orthorhombic (γ-phase) CsPbI3, suggesting the beneficial substitution of a small quantity of DMA+.As a result, the PSC exhibits a satisfactory PCE of 19.76%, while maintains high stability (95% retention for ~186 h by MPP tracking test).
Designing suitable Dion–Jacobson (DJ)/hybrid perovskite, including 4-aminopiperidinium-based quasi-DJ-2D perovskite74, amidino-based DJ-2D perovskite96, utilization of suitable solvent and ligand for quasi-2D-DJ phases on 3D perovskites142, and fabrication of organic-inorganic hybrid perovskite by incorporating a tiny amount of DMA+ for Cs+ 143 enhanced the efficiency and/or stability of the PSCs.
Strategies for using ambient conditions and/or water for the fabrication of PSCs
The fabrication of PSCs in ambient air and/or water can prevent the moisture-induced severe degradation of the perovskite layer, and that can trigger their industrialization. The introduction of guanabenz acetate salt can inhibit perovskite hydration, alleviate both anion and cation vacancies, and crystallize a high-quality film for the fabrication of PSCs in ambient air. Yan et al.71 have utilized guanabenz acetate salt to prevent perovskite hydration, obviate both anion and cation vacancies, and crystallize a high-quality film for the fabrication of PSCs in ambient air. Hence, the PSC exhibits a remarkable PCE of 25.32% (Table 5), while it exhibits high stability (96% retention for 500 h by MPP tracking test; 85% retention for 300 h under damp heat conditions with 85% relative humidity at 85 °C). The cause of the generation of good-quality perovskite sheet in ambient air was determined. There is an initial interaction stage of moisture with perovskite, but the hydration pathway is blocked by the utilization of guanabenz acetate salt, and thus a high-quality perovskite film is formed in ambient air. The left side of Fig. 6a depicts the mechanism of moisture-induced decomposition. The water molecules can be more easily bound in energetics with an atom exposed by a vacancy in perovskite at the initial stage of the hydration process. Consequently, FAI will be escaped and the Pb2I62− framework will be collapsed. The DFT calculations disclose the binding energy of water molecules with the perovskite (Fig. 6b) and the critical role of vacancies on hydration. FA+ and I− vacancies are readily generated during the deposition of the film. If I− vacancy occurs, then the binding energies are −1.450 eV for H2O with FA+, − 0.610 eV for H2O with Pb2+, and −1.189 eV for H2O with I−. If FA+ vacancy occurs, then the binding energies are −1.134 eV for H2O with FA+, −1.133 eV for H2O with Pb2+, and −1.461 eV for H2O with I−. However, in the ideal perovskite structure, the binding energies are 1.020 eV for H2O with FA+, 1.446 eV for H2O with Pb2+, and 1.774 eV for H2O with I−. These results disclose that the degradation of the perovskite can occur due to the hydration of the perovskite by the absorption of water molecules at the sites adjacent to vacancy defects. Thus, eliminating vacancies can be a viable way to diminish the sensitivity of perovskites to moisture.
a Schematic diagram illustrates the moisture-induced decomposition of perovskite film (left side) where the addition of GBA reduces the moisture sensitivity (right side); b The binding energies of H2O molecules due to the interaction with perovskite via H2O-I−, H2O-Pb2+, and H2O-FA+ approaches when the structure contained no vacancies, I− vacancies, and FA+ vacancies; c FTIR spectrum of GBA in comparison with the target film; d High resolution XPS 4f spectrum of Pb obtained from control film in comparison with target film; e Deep energy level transient spectroscopy (DLTS) spectrum of target film in comparison with control film, and f corresponding Arrhenius plots with fitting, where the data points in (f) are acquired through calculating internal transients, which have been included in the DLTS signals via the discrete Laplace transform, while the lines are linear fits of the data points; g The binding energies of H2O molecules due to the interaction with the perovskite via H2O-I−, H2O-Pb2+ and H2O-FA+ approaches when the perovskite film incorporated with GBA (reproduced with permission from ref. 71 Copyright 2023, Springer Nature).
The right side of Fig. 6a depicts that the utilization of guanabenz acetate salt can remove vacancies through interaction with perovskite. The aromatic amine salt guanabenz acetate contains acetate (AC−) and guanidine groups, where the AC− exhibits robust coordination ability, while a larger number of effective hydrogen bonds can be formed by the terminated guanidine group in the benzene ring when compared to that of FA+. In the FTIR spectra (Fig. 6c), the peaks of C=O and C-O of guanabenz acetate salt with perovskite film exhibit a shift towards a lower wave number when compared to that of guanabenz acetate salt, suggesting the existence of an interaction of guanabenz acetate salt with perovskite. Furthermore, in the high-resolution XPS spectra of Pb 4f (Fig. 6d), the target film affords an obvious downshift when compared to its counterpart, which suggests that the guanabenz acetate salt can bind with the unsaturated Pb2+, thereby eliminating the I− vacancy. If I− vacancies occur, the unsaturated Pb2+ will preferentially bind with AC− by a robust Pb-O bond instead of the water molecule because the binding energy of Pb2+-AC− is −2.401 eV, which is much lower than that of the −0.610 eV for Pb2+-H2O. Furthermore, the energetic stability calculation and generated hydrogen bond suggest that the terminated guanidine group of guanabenz acetate salt can be responsible for the filling of FA+ vacancies. In the high-resolution XPS spectra, the target film exhibits a downshift when compared to that of the control film, which suggests that the AC− is responsible for the filling of the I− vacancy and the generation of a hydrogen bond between the I atom and the terminated guanidine group of guanabenz acetate salt. In the FTIR spectra, the peak of C=N of guanabenz acetate salt with PbI2 film exhibits a shift towards a lower wave number when compared to that of guanabenz acetate salt, suggesting the existence of an interaction of the terminated guanidine group of guanabenz acetate salt with PbI2 film by a hydrogen bond. Additionally, the DFT calculations suggest that the formation of I− vacancy is suppressed by 0.28 eV when FA+ vacancy is filled by the terminated guanidine, and thus a stable perovskite lattice is formed.
A deep energy level transient spectroscopy (DLTS) investigation (Fig. 6e) is performed to validate the elimination of I− and FA+ vacancies by guanabenz acetate salt in perovskite. In the DLTS plot, the control sheet shows two peaks indicated as T1 and T2, suggesting the existence of two kinds of dominant traps. In contrast, the absence of trap peaks is observed in the target film, suggesting the elimination of vacancy after the introduction of guanabenz acetate salt in perovskite. To determine the activation energy of the corresponding traps, the Arrhenius plots have been fitted (Fig. 6f) based on the Arrhenius equation (ln(tau × Vth × Nc) = ((ET − EV)/KT) − ln(XPσP)). The symbols denote the following terms: K: Boltzmann constant; Nc: effective density of states of the trap in cm−3; XP: entropy factor; tau: emission time constant; Vth: thermal velocity; ET: energy level of the trap center; EV: energy level of VBM; σP: capture cross section; T: temperature of the test. The ΔE = ET − EV is the trap activation energy, where the ΔE of T1 is calculated to be 0.51 eV for FA+ vacancy and the ΔE of T2 is calculated to be 0.39 eV for I− vacancy in the control film. The space-charge-limited current investigation further corroborates the DLTS investigation. Furthermore, the target film exhibits higher carrier mobility (2.68 cm2 V−1 s−1) and diffusion length (3.92 μm) when compared to that of the control film (1.98 cm2 V−1 s−1 and 1.96 μm). Additionally, the effects of the diminished moisture sensitivity of perovskite with guanabenz acetate salt due to the elimination of vacancies are analyzed. The FA+ vacancy is filled by terminated guanidine groups, and the I− vacancy is filled by AC− of guanabenz acetate salt, where the calculated binding energies (Fig. 6g) are 0.790 eV for H2O molecules with FA+, 0.373 eV for H2O molecules with Pb2+, and 0.475 eV for H2O molecules with I−. The binding energies of H2O molecules with the perovskite are negative (Fig. 6b), whereas the binding energies of H2O molecules are positive (Fig. 6g) after the introduction of guanabenz acetate salt, which suggests that the H2O molecules are harder to bind (not spontaneous or immediate) with the perovskite after the introduction of guanabenz acetate salt. Therefore, the perovskite hydration is intercepted due to guanabenz acetate salt, which could guard the crystallization of perovskite from moisture, and thus high-quality perovskite film deposition is obtained in ambient air (Fig. 6a) for the fabrication of efficient PSCs.
The introduction of dry air and a surfactant (potassium oleate) can enable water to act as a green solvent for the fabrication of perovskite. Zhang et al.72 have utilized dry air and a surfactant (potassium oleate), which enables water to act as a green solvent for the fabrication of perovskite, to achieve enhanced performance of PSCs. At first, the crystallization process of Pb(NO3)2 under various relative humidity conditions is investigated. The moisture in the ambient air acts a crucial role in the nucleation rate of Pb(NO3)2 and the spreading dynamics of aqueous inks, where the high surface tension of water can severely deteriorate the morphology of the perovskite film. Hence, the surface tension of water is diminished by using long-alkyl-chain potassium oleate as an anionic surfactant. Therefore, pinhole-free, high-quality, conformal perovskite film with enlarged grain size can be prepared by the synergistic effect of oleate anion and dry air, while potassium oleate modulates the crystallinity of Pb(NO3)2, which can improve the crystallization of PbI2 and enhance the formation kinetics of perovskite. As a result, the PSC exhibits a high PCE of 24.14%, while it exhibits high stability (95.2% retention for 800 h by MPP tracking test).
The introduction of NH4+ as a volatile cation can easily eliminate acetate during annealing for the fabrication of high quality perovskite film in an ambient laboratory environment. Zhao et al.144 have utilized NH4+ as a volatile cation for the facile elimination of acetate during annealing, and thus high-quality formamidinium-caesium mixed-cation perovskite films are prepared using a lead acetate-based precursor formulation by blade-coating in an ambient laboratory environment. As a result, the PSC and module exhibit high PCE of 21% and 18.8% (10 cm2), respectively, while the PSC exhibits high stability (80% retention for 327 h by MPP tracking test (PTAA as HTL)) and high thermal stability (negligible decay for 3300 h at 65 °C).
Utilization of machine learning-guided robotic platforms can enable the fabrication of high quality perovskite in an ambient atmosphere. Zhang et al.145 have demonstrated the SPINBOT (fully automated spin-coating platform) for the engineering of solution-processed functional thin films for high-performance PSCs. Using SPINBOT, experiments can be conducted with high sampling variability, where hundreds of substrates can be processed without supervision with superior experimental control. The Bayesian optimization algorithm can enable the iterative optimization process, and thus an intricate parameter space can be explored by the SPINBOT, which constantly enhances the reproducibility and quality of the prepared thin films. Thus, the exploration of the optimization process of PSCs can be enabled by this machine-learning-guided reliable SPINBOT platform through the facile photoluminescence characterization of thin films. Thus, utilizing a machine-learning-guided, robotic platform, the perovskite thin-film parameter spaces are optimized. As a result, the PSC obtained in an ambient atmosphere exhibits a high PCE of 21.6%, while it exhibits high stability (85% retention for 1570 h by MPP tracking test).
Utilization of aqueous halide-free nanofluids can control the formation kinetics of a perovskite through a grain refinement strategy for the fabrication of green water-processed PSCs. Zhai et al.146 have utilized aqueous halide-free nanofluids to control the formation kinetics of a perovskite through a grain refinement strategy, while in situ UV-Vis absorption and PL measurements are performed to investigate the crystallization process, where anion configurations and the microstructure of PbI2 play a vital role in the formation kinetics. Subsequently, the reaction rate is promoted to ∼30% by using PbCO3 nanofluids with an inert CO32− (carbonate anion), where the formation of FA vacancy defects is efficiently hindered by CO32− at the surface of a FA-based perovskite. As a result, the green water-processed PSC obtained in dry-air atmosphere exhibits a high PCE of 23.64% while it exhibits high stability.
Strategies for using ambient conditions and/or water for the fabrication of PSCs, including the utilization of guanabenz acetate salt71, dry air and surfactant (potassium oleate)72, NH4+ as a volatile cation for the facile elimination of acetate during annealing144, machine learning-guided robotic platforms145, and aqueous halide-free nanofluids146, enhanced the efficiency and/or stability of the PSCs.
Flexible/bifacial PSCs
Developing flexible PSCs with high performance is highly desirable for the application of wearable electronics, intelligent vehicles, uncrewed aerial vehicles, and building-integrated photovoltaics76,147. Utilization of molecular dipole engineering strategy can release the strain for the fabrication of mechanically strong and flexible PSCs. Xie et al.76 have fabricated mechanically strong and flexible PSCs by using molecular dipole engineering strategy to release the strain. 1F-2CN, 2F-2CN, and 3F-2CN are among the -CN additives with different molecular dipoles that are created (1F-2CN: 2’-fluoro-[1,1’-biphenyl]-3,5-dicarbonitrile; 2F-2CN: 2’,6’-difluoro-[1,1’-biphenyl]-3,5-dicarbonitrile; 3F-2CN: 2’,3’,4’-trifluoro-[1,1’-biphenyl]-3,5-dicarbonitrile). Two -CN groups in the additives may coordinate the Pb2+ defects, and the F (fluorine) atoms may control the dipole moment of the additives by forming hydrogen bonds with the charged FA+ group. Additionally, the -CN additions may stitch the flaws at the grain boundaries and relieve the stress at the grain boundary, resulting in a lower Young’s modulus and greater mechanical flexibility. Furthermore, the additions may improve carrier extraction and transport while attenuating the interactions between the longitudinal optical phonons and the charge carriers. When compared to PSC with 1F-2CN and PSC with 3F-2CN, the flexible PSC with the 2F-2CN addition shows superior efficiency and stability due to its stronger molecular dipole. Thus, the PSC containing the 2F-2CN addition shows a high PCE of 24.08% (Table 6), strong stability (retention of 94% for 620 h by MPP tracking test), and excellent bending cycle stability for flexible solar cells (92% retention for 3000 bending cycles).
The utilization of halide-substituted ammonium salts can optimize the buried interface and the perovskite crystallization, eliminate the nanovoids at the interface for the fabrication of flexible PSCs on flexible plastic substrates. An et al.148 have utilized halide-substituted ammonium salts to optimize the buried interface and the perovskite crystallization to enhance the performance of flexible PSCs, where the halide-substituted ammonium salts are n-bromophenethylammonium (n-Br-PEAX; n = 2 or 4; X = Cl or Br). The speed of the crystallization process of perovskite can be altered by the rational design molecular structure of the ammonium salt, thereby generating a dense and homogenous morphology where nanovoids are absent at the interface. Hence, the PSC exhibits a high PCE of 20.2% on flexible plastic substrates, while it exhibits high stability (87% retention for 290 h by MPP tracking test).
The utilization of in situ crosslinking-assisted perovskite grain growth strategy can enable the fabrication of mechanically robust flexible PSCs with high efficiency. Wu et al.149 have demonstrated mechanically robust flexible PSCs with high efficiency through in situ crosslinking-assisted perovskite grain growth. The bis((3-methyloxetan-3-yl) methyl) thiophene-2,5-dicarboxylate (OETC) monomer is designed for regulating the growth of perovskite on plastic substrates. Thus, a perovskite film with a preferential crystal orientation, compact stacking, and enlarged grain size is obtained. Additionally, the mechanical stress can be effectively removed by the crosslinked elastomer polymer accumulated at the grain boundaries of the perovskite. Thus, after 2000 bending cycles (Radius: 5 mm), the SEM image of perovskite-crosslinking OETC film exhibits negligible fracture (Fig. 7c), but the SEM image of perovskite-MOTC film exhibits noticeable fracture (Fig. 7b), whereas the SEM image of pristine perovskite film exhibits severe fracture (Fig. 7a), suggesting the crosslinking OETC affords robust deformation stability, while the MOTC affords a partial deformation stability, but the pristine perovskite possesses poor deformation stability (MOTC: bis(3-methoxypropyl) thiophene-2,5-dicarboxylate). As a result, the PSC with crosslinking OETC exhibits high bending cycle stability for flexible solar cells (93% retention for 5000 bending cycles (Fig. 7d)), and the PSC with crosslinking OETC exhibits high PCE of 23.4%, while it exhibits high stability.
Top-view SEM images of a pristine, b MOTC, and c COETC-based perovskite films after 2000 bending cycles (Radius: 5 mm); d PCE of the flexible PSCs versus bending cycles (Radius: 5 mm) (reproduced with permission from ref. 149 Copyright 2022, Elsevier Inc.). The performance of the bifacial PSC with concurrent front and back illumination: e Schematic diagram illustrates the setup for measurement for bifacial illumination through two simulators; f J–V curves with forward (fwd) and reverse (rev) scans; and g corresponding steady-state equivalent efficiency or stable power output (SPO) of the bifacial PSCs with concurrent front and back bifacial illumination, under the 1-sun intensity from the front side with 100 mW/cm2, and several adjustable light intensities from the rear side with 0–50 mW/cm2 (albedos: 0–0.5) (reproduced with permission from ref. 77 Copyright 2023, Elsevier Inc.).
Bifacial PSCs can harvest more solar illumination through two sides (front and rear surfaces; additional energy can be added through albedo light) than the one-side light harvesting of the monofacial PSCs, and that can enhance the energy conversion efficiency of the bifacial PSCs. Nevertheless, the design of the bifacial PSCs with high PCE and stability is a challenging task because of electrode corrosion and electrical loss in transparent rear contact, ion migration, and high rear side carrier recombination, while the components (such as the back contact electrode, perovskite layer, and transport layer) of the bifacial solar cells (making these as semi-transparent PSCs) need to be transparent to achieve high performance150,151. Thus, Jiang et al.77 have fabricated a transparent conducting rear electrode for bifacial single-junction PSCs (Fig. 7e) with inverted architecture using optical and electrical modeling to achieve high efficiency at different albedo illumination conditions. They have demonstrated that the bifacial PSCs have the capability to surpass their monofacial PSCs in terms of a lower levelized cost of energy and larger energy yields. As a result, the single-junction bifacial PSC exhibits a high PCE of 31.2% at an albedo of 0.5 (Fig. 7f), and the stabilized bifacial power output densities are 26.9 mW/cm2 under an albedo of 0.2, 28.5 mW/cm2 under albedo of 0.3, and 30.1 mW/cm2 under an albedo of 0.5 (Fig. 7g), while the bifacial PSCs exhibit the bifaciality of about 91% to 93%.
Various strategies for flexible PSCs, including molecular dipole engineering-assisted strain release76, utilization of halide-substituted ammonium salts148, and in situ crosslinking-assisted perovskite grain growth149, enhanced the efficiency and/or stability of the PSCs. Fabrication of a transparent conducting rear electrode77 enhanced the efficiency and/or stability of the bifacial PSCs.
Additive strategies for PSCs
Utilizing ammonium cations with a high pKa value of 12 as an additive can inhibit the deprotonation process when they react with FA+ in the perovskites at high-temperatures light soaking, and that can enhance the efficiency and stability of the PSCs at high-temperatures. Wang et al.63 have observed that the extensively used organic cations such as butylammonium (BA+) and phenethylammonium (PEA+) for 2D perovskites are unstable in formamidinium (FA+)-containing perovskites in light soaking at high-temperature. The BA+ and PEA+ deprotonate into amines at high-temperature light soaking, which subsequently react rapidly with FA+ to generate (butylamino)methaniminium (BAMA+) and (phenethylamino)methaniminium (PEAMA+), respectively. As a result, ammonia is produced, which generates damage to grain boundaries and perovskite interfaces and hampers the photostability of the PSCs at high-temperatures. Therefore, the photostability of the PSCs at high-temperatures can be enhanced by either removing BA+ and PEA+ cations from precursors or utilizing ammonium cations with high pKa (12.0), including BAMA+ and PEAMA+, because of the difficulty in deprotonation. Thus, the PSC exhibits a high PCE of 24.1% (Tables 7), while it exhibits high stability (90% retention at 85 °C for ~1500 h) by MPP tracking test.
The cation compositional inhomogeneity exhibited due to A-site cation segregation can hamper the photovoltaic performance of the PSCs with FA1-xCsxPbI3. The utilization of a suitable additive with a sulfone group can cause cation compositional homogeneity, and that could enhance the performance of PSCs. Liang et al.80 have identified the cause of the compositional inhomogeneity outside of the plane and its significant impact on PSCs. They envision this occurring along the vertical direction throughout perovskite films, where Cs aggregation is observed, preferably at the bottom of the film (Fig. 8a). Furthermore, the addition of 1-(phenylsulfonyl)pyrrole (PSP; Fig. 8b) with a sulfone group homogenizes the distribution of cation content in perovskite films. As a result, the PSC shows a high PCE of 26.09% and a good level of stability (92% retention for 2500 h) according to the MPP tracking test. In situ synchrotron radiation grazing incidence wide-angle X-ray scattering (GIWAXS) is conducted to determine the dual essential kinetic processes of phase transition and crystallization during the formation of perovskite. As shown in Fig. 8c, signals for a q vector of about 0.8 Å−1 can be ascribed to δ-phase perovskite of 2H (100), at about 0.82 Å−1 can be attributed to δ-phase perovskite of 6H (101), and 1.0 Å−1 can be assigned to α-phase perovskite. Two periods can be defined to investigate the kinetic processes. Period I is ascribed until the appearance of α-phase perovskite from the dripping of chlorobenzene (CB), where the crystallization rate is indicated by the duration of period I. Period II indicates the transition rate of δ- to α-phase, signifying the duration of stable α-phase formation. Thus, the in situ GIWAXS results disclose that both the phase transition and crystallization are accelerated by the introduction of PSP. The ‘out-of-plane cation inhomogeneity’ studies and in situ GIWAXS results disclose that the segregation of the Cs and FA phases can be efficiently inhibited by the PSP, where the slow rate of phase transition and crystallization are the potential kinetic factors leading to segregation of phase. The barrier energy (EB) for the perovskite phase change and crystallization is calculated thermodynamically using DFT calculations (DFT: Density functional theory), and the energy is then divided into the contributions from the Cs and FA components. The ΔEB = EBFA – EBCs defines the change in barrier energies. The mismatch factor μ = (EBFA – EBCs)/EBFA is calculated to determine more precisely the imbalance between the Cs and FA components. During period I, the ΔEB,I ref is 101.6 meV for the reference system (Fig. 8d), whereas the ΔEB,I PSP is only 35.3 meV for the PSP system (Fig. 8e), while the corresponding μ values are calculated to be 20.48% for μI,ref and only 5.34% for μI,PSP. At period II, ΔEB,II ref is 82 meV with μref is 12.49% for the reference system, but ΔEB,II PSP is only −6 meV with μPSP is only −1.79% for the PSP system. Thus, the PSP system exhibits lower μI and μII, which disclose that the variations in the phase transition and crystallization rates of the Cs and FA components are diminished, where the differences in the cations rates are possibly attributed to the observed delay through in situ GIWAXS. The potential reason for cation inhomogeneity may be attributed to the soft base characteristic of Cs cations compared to FA cations. This difference could lead to stronger interactions with PbI3−, resulting in a tendency for Cs to aggregate preferentially at the bottom. Moreover, the variation in the solubilities of FA and Cs species could partially be ascribed to inhomogeneity of cations.
a Schematic diagram depicts the spatial vertical segregation of the FA and Cs phases in FA-Cs perovskite, where inhomogeneous phase distribution is caused by out-of-plane FA and Cs segregation; b Electrostatic potential image with molecular structure of 1-(phenylsulfonyl)pyrrole (PSP) additive, where the PSP can retard the segregation of cations in FA-Cs perovskite; c–i Disclosing the origin of the segregation of the Cs and FA phases: c In situ GIWAXS pattern of PSP system in comparison with reference system, disclosing the processes of crystallization and phase transition, where the color bars range from 0 to 1; Schematic diagrams depict the computation results for free energy evolution in the d reference system in comparison with e PSP system during crystallization and the phase transition processes; The blue rectangle indicates the relevant FAPbI3 phases, while the red rectangle represents the relevant CsPbI3 phases; The blue solid line represents the free energy evolution of FAPbI3, while the red solid line indicates the free energy evolution of CsPbI3; Plots depict the Fourier-transformed R space results obtained from EXAFS measurements of the f reference films and g PSP films, where the dashed lines at 2.9 Å correspond to the Pb–I coordination, and 2.2 Å correspond to the Pb–O coordination; h Pb–O coordination ratios obtained from the EXAFS measurements; i FTIR spectra of PSP in comparison with PSP(PbI2) complex (reproduced with permission from ref. 80 Copyright 2023, Springer Nature).
Through extended X-ray absorption fine spectroscopy (EXAFS), the adsorption spectra of the Pb LIII edge are obtained to determine the interactions of PSP with perovskite. A total of five grazing incidence angles are chosen in order to collect data at various perovskite film depths. The peaks at radial distances of about 2.2 Å and 2.9 Å, respectively, can be attributed to Pb-O and Pb-I coordination, as seen in Fig. 8f, g. A gradual downward shift of about 0.03 Å for the coordination of Pb-I is seen for the reference film (Fig. 8f) as the detecting depth is increased, suggesting that the lattice at the bottom of the perovskite has compressed. Nonetheless, the comparatively steady peaks linked to Pb-I coordination are detected subsequently to the addition of PSP, indicating the existence of cation inhomogeneity outside of the plane. When the depth is increased, an obvious shift in the higher radial distance for the Pb-O coordination is observed for the PSP film (Fig. 8g), suggesting the formation of a higher Pb-O coordination near the bottom of the film. Calculating the Pb-O coordination ratio of (Pb-O)/((Pb-O) + (Pb-I)) (Fig. 8h) reveals that Pb atoms in the PSP system typically coordinate with extra oxygen atoms toward the bottom of the perovskite film. Thus, it is hypothesized that there is a possible interaction between PSP and Pb atoms in perovskite due to the donation of electrons from the two oxygen atoms of the PSP.
Fourier transform infrared spectroscopy (FTIR) measurements are performed on the as-synthesized (PbI2)x(PSP)y complex crystals to exactly determine the interaction of PSP with PbI2, where the peak at about 1328 cm−1 corresponds to asymmetric stretching vibration (νas), 1133 cm−1 corresponds to symmetric stretching vibration (νs) of the sulfone (O = S = O) group, and 964 cm−1 corresponds to symmetric stretching vibration (νs) of the sulfoxide (S = O) group. Moreover, all three characteristic peaks exhibit obvious shifts, suggesting the coordination between PSP and PbI2 by the sulfone (O = S = O) group (Fig. 8i). The νas and νs peaks exhibit upward shifts, which suggest that both oxygen atoms of the PSP can act as active sites, while the coordination between the O = S = O group and PbI2 is further confirmed by nuclear magnetic resonance (NMR) spectra, where the shifts in the carbon atoms nearby to the O = S = O group (nos. 14, 11, 6, 2, and 1) to a higher field are observed. These results corroborate the peak shifts detected through XPS measurements and the diminished PbI2 signal obtained through in situ GIWAXS tests.
Utilization of a suitable Lewis base additive can enhance the crystallinity and diminish the defect density and internal residual stress of the perovskite bulk, and that can enhance the performance of the PSCs. The quality of the bulk and surface of the FA0.98Cs0.02PbI3 perovskite film was improved by Chen et al.152 to diminish the efficiency gap. At first, a Lewis base additive (dibutyl sulfoxide) is used to enhance the crystallinity and diminish the defect density and internal residual stress of the perovskite bulk. Finally, the surface of the perovskite film is treated with trifluorocarbon-modified phenethylammonium iodide to optimize the energy levels and protect the film from moisture and passivate defects. Thus, the PSC exhibits a high PCE of 25.1%, while it exhibits high stability (97.4% retention for 1800 h) according to the MPP tracking test.
The introduction of a non-volatile, multifunctional additive can alter the kinetics of the growth of the perovskite film via a hydrogen-bond-bridged intermediate phase, and that can enhance the performance of the PSCs. Li et al.153 have utilized a non-volatile, multifunctional additive to alter the kinetics of the growth of the perovskite film via a hydrogen-bond-bridged intermediate phase. Thus, coherent grain growth and the generation of large perovskite grains from the bottom to the surface of the perovskite film are achieved, leading to considerably diminished non-radiative recombinations. As a result, the PSC exhibits a high PCE of 24.8% and a low energy loss of 0.36 eV, while it exhibits high stability (90% retention for 500 h) by MPP tracking test, while the unencapsulated devices afford enhanced thermal stability with a T98 lifetime for >1000 h in a nitrogen-filled glovebox under constant heating at 65 ± 5 °C.
The introduction of dimethylammonium chloride as an additive can modulate the perovskite intermediate precursor phases, which can modulate the grain size, orientation, crystallinity, and texturing of the perovskite system, and that can enhance the performance of the PSCs. McMeekin et al.154 utilized dimethylammonium chloride as an additive to modulate the perovskite intermediate precursor phases through a high-temperature dimethyl-sulfoxide-free processing route. Thus, the grain size, orientation (corner-up versus face-up), crystallinity, and texturing of the (FA)yCs1–yPb(IxBr1–x)3 perovskite system are modulated through controlling the sequence of crystallization. Hence, the PSC exhibits an unexpected PCE of 20.2% while it exhibits high stability (it exhibits 80% retention for 1190 h for steady-state power conversion efficiency and 80% retention for 1410 h at open-circuit conditions in air under simulated sunlight at 65 °C).
Excess lead iodide (PbI2) and abundant defects are commonly observed in perovskite films, where the PbI2 species can exhibit either a positive or negative effect on the PCE of the PSCs. The PbI2 layer with an ordered array structure can be obtained by integrating a suitable additive with nanoimprinting technology, and that can enhance the performance of the PSCs. He et al.155 have integrated a succinamide additive with nanoimprinting technology for the fabrication of a porous PbI2 layer with an ordered array structure. Thus, organic amine salts can be confined in PbI2 arrays to enhance omnidirectional diffusion and realize adequate conversion to perovskites. As a result, the PSC exhibits a high PCE of 23.56% and high stability, while the module (14.63 cm2) exhibits a PCE of 16.42%.
The introduction of more hydrophobic π-conjugated aromatic additives can enhance the passivation and decrease the recombination, which can improve the stability and PCE of PSCs. Wang et al.156 have demonstrated the role of π-conjugated aromatic additives (CAAs) such as coumarin methacrylate (CMA) and coumarin hydroxyethyl (CHE) on the stability and PCE of PSCs using Cs0.05(FA0.85MA0.15)0.95Pb(I0.85Br0.15)3 perovskite. The additives are added to the precursor perovskite solutions, where CMA is more hydrophobic than CHE. Thus, the extent of passivation and binding to Pb2+ increase with an increase in hydrophobicity, leading to a decrease in recombination. As a result, the PSC with CMA exhibits a superior PCE of 21.28% than that of CHE (21.14%), while it exhibits high stability (including moisture and thermal stability).
The introduction of Zn(C6F5)2 as a molecular additive, and the utilization of dopant-free hole transport layer can enhance the performance of the PSCs. Mali et al.157 have demonstrated β–CsPbI3-Zn(C6F5)2 and γ–CsPbI3-GAI based phase-heterojunction all-inorganic PSC (n-i-p type) with high efficiency, where the β–CsPbI3-Zn(C6F5)2 was obtained by hot-air-processed and dimethylammonium iodide-assisted method, while the γ–CsPbI3-GAI was obtained by triple-source thermally evaporated method (GAI: guanidinium iodide). The Zn(C6F5)2 is used as a molecular additive, while the dopant-free hole transport layer is utilized. Therefore, the β − γ-based phase-heterojunction PSC and module exhibit enhanced PCE of 21.59% and 18.43% (18.08 cm2), respectively, while the PSC exhibits high stability (~90% retention for 200 h by MPP tracking test).
Utilization of hydrophobic additive with the hole transport layer can enhance the moisture resistance of the perovskite films, while integration of silica nanoparticles with suitable size and spacing with the perovskite films can retrieve the absorption loss induced by the lack of reflective metal electrodes, and that can enhance the performance of the bifacial perovskite minimodules. It has been shown by Gu et al.158 that the bifacial perovskite minimodules can add additional energy through albedo light, and their front efficiency is comparable to that of their opaque monofacial counterparts. A hydrophobic additive is introduced with the hole transport layer to enhance the moisture resistance of the perovskite films, while silica nanoparticles with suitable size and spacing are integrated with the perovskite films to retrieve the absorption loss induced by the lack of reflective metal electrodes. Under 1 solar illumination and 0.2 albedo, the small-area single-junction bifacial PSCs show a power-generation density of 26.4 mW cm−2. As a result, the PSC exhibits a high PCE of 23.19% while it exhibits high stability. The bifacial minimodules exhibit >20% front efficiency, 74.3% bifaciality, and >23 mW cm−2 of power-generation density at an albedo of 0.2. The bifacial minimodule exhibits high stability (97% retention for >6000 h after light soaking under 1 sun at 60 ± 5 °C).
Utilization of suitable alkylthiol additive with the hole transport material can enhance the performance of the PSCs. Liu et al.159 have demonstrated a facile approach to introducing the 1-dodecanethiol (DDT, a low-cost alkylthiol additive) in the hole transport material (spiro-OMeTAD: 2,20,7,70-tetrakis(N,N-di-p-methoxyphenyl-amine)-9,90-spiro bifluorene). Thus, a controllable, efficient doping process with efficient doping duration is achieved due to the DDT, which can provide comparable performance for hole transport material before air activation. The coordination of DDT with a lithium compound (LiTFSI) elevates the concentration of dopants in the bulk hole transport material, diminishes their accumulation at interfaces, and improves the structural integrity of the hole transport material at heat, wetting, and light stress. Consequently, the PSC demonstrates substantial power conversion efficiency (PCE) of 24.6% and maintains robust stability, retaining 90% of its performance after 1000 h as verified by the maximum power point (MPP) tracking test.
The utilization of suitable additives, including additives with a sulfone group for perovskite80, Lewis base additives for perovskite152, a non-volatile, multifunctional additive for perovskite153, dimethylammonium chloride as an additive for perovskite154, the integration of succinamide additives with a nanoimprinting technology for perovskite155, π-conjugated aromatic additives for perovskite156, Zn(C6F5)2 as a molecular additive157, and the introduction of 1-dodecanethiol as an alkylthiol additive with the hole transport material159 enhanced the efficiency and/or stability of the PSCs.
Strategies associated with buried interfaces for PSCs
The main obstacles hindering the photovoltaic stability and performance of inverted PSCs are photochemical degradation and non-radiative recombination losses (trap-assisted) at the perovskite and charge-transport layers interface. Introduction of suitable additive into the precursor inks can tune the crystallization of perovskite, regulate the extraction dynamics and carrier recombination at the buried interface, and that can enhance the performance of the PSCs. Li et al.64 have introduced a tiny quantity of 2-aminoindan hydrochloride into the precursor inks to tune the crystallization of methylammonium-free perovskite. Thus, the additive regulates the extraction dynamics and carrier recombination at the buried interface through the generation of a bottom-up 2D/3D heterojunction (Fig. 9a, b). The PSC achieves a notable PCE of 25.12% overall (Fig. 9a, b) and a certified PCE of 24.60%, surpassing previously reported values for similar MA-free inverted PSCs (Fig. 9c). It maintains over 98% of its performance after 1500 h, as verified by MPP tracking tests (Table 8 and Fig. 9e).
a Schematic diagram depicts the configuration of the inverted PSC device having bottom-up 2D/3D perovskite heterojunction at the buried interface; b HRTEM image (Cross sectional view) of the target PSC; The inset (3D perovskite) depicts the enlarged TEM image of the area enclosed by a yellow box; The formed 2D perovskite is indicated by the areas enclosed by the dashed white lines; The lattice parameters for 3D and 2D perovskites are provided; c The PCE of the target inverted PSC in comparison with the PCEs (>22.8%) of the reported PSCs (Ref. 4, 198, Ref. 6, 199, Ref. 7, 200, Ref. 8, 201, Ref. 11, 202, Ref. 12, 203, Ref. 14, 204, Ref. 15, 205, Ref. 16, 206, and Ref. 55, 207). d J–V curve of the target PSC device (Area: 1 cm2), where the inset depicts its photovoltaic parameters; e PCE of the unencapsulated target devices in comparison with the control device obtained for 1500 h at maximum power point under N2 atmosphere at 55 ± 5 °C under continuous one-sun illumination with the initial PCE of 21.73% (control), 24.42% (target-1) and 24.11% (target-2) (reproduced with permission from ref. 64 Copyright 2023, Springer Nature).
Introduction of an amphiphilic molecular hole transporter with multifunctional group can produce a superwetting underlayer for the deposition of perovskite, and that can cause the formation of high-quality perovskite films with the fewest defects at the buried interface. Zhang et al.81 have demonstrated the (2-(4-(bis(4-methoxyphenyl)amino)phenyl)-1-cyanovinyl)phosphonic acid (MPACPA) containing a multifunctional cyanovinyl phosphonic acid group as an amphiphilic molecular hole transporter. This generates a superwetting underlayer for the deposition of perovskite, and that causes the formation of high-quality perovskite films with the fewest defects at the buried interface. As a result, the perovskite film exhibits 17% of photoluminescence quantum yield and about 7 microseconds of Shockley-Read-Hall lifetime, and the perovskite solar cell and minimodule exhibit high PCE of 25.16% (23.4% for 1 cm2) and 22.0% (10 cm2), respectively. The PSC exhibits robust stability (>90% retention for 2000 h) by MPP tracking test due to the encapsulated modules afford high stability at damp heat and operational test conditions. Electronic structure calculations by first-principles are conducted to determine the effect of passivation of MPACPA on typical deep-level defects generated on the surface of the perovskite grain, including lead-iodide antisite (PbI) and interstitial lead (Pbi) defects. Deep defective states are induced by Pbi and PbI within the band gap, and that could play as non-radiative recombination centers. The introduction of the MPACPA molecule significantly passivates the defective states of PbI and Pbi, causing them to migrate either within the conduction or valence bands or close to the band edges, as shown in Fig. 10b. Emerging chemical bonds were generated between N and Pb from the cyano group (Pb-N’) and between O and Pb from the phosphonic acid group (Pb-O’), as shown in Fig. 10a, leading to the formation of a complemented octahedral local chemical environment for Pb. The Pb-N’ bond length is calculated to be 2.47 Å, which is less than that of the experimentally determined Pb-N bond lengths (~2.58 to 2.75 Å) in lead acesulfamates, whereas the calculated Pb-O’ bond length of 2.68 Å is within the experimentally determined limit of ~2.484 to 2.914 Å. The calculated electron localization function results further confirm the chemical bond formation between Pb and O’ and N’ (Fig. 10c).
a The passivated surface with optimized structure (Pbi: interstitial Pb), and b the density of states (DOS) of the defective and the passivated surfaces, where the DOS is projected onto individual atoms: I and Pb atoms in the perovskite, a particular N’ atom in the cyano group, a particular O’ atom forming the P = O in the phosphonic acid group, where the energy of the valence band maximum has been set to zero; c The calculated electron localization function in the area of the passivated molecular configuration and the defective molecular configuration; d The apparent trap density of states acquired from thermal admittance spectroscopy for devices with various HTLs (reproduced with permission from ref. 81 Copyright 2023, American Association for the Advancement of Science).
The synergistic effect of passivation through two types of bonds significantly enhances the thermodynamic stability of passivation sites, proving especially effective at mitigating deep-level defect states. As a result, the perovskite film on MPACPA exhibits 17% of the photoluminescence quantum yield in the half-stacks due to the decrease in non-radiative recombination at the buried interface. The XPS studies further confirm the interaction of CPA with Pb. The thermal admittance spectroscopy studies disclose the frequency dependent capacitance in PSCs with various hole transport layers. The MPACPA-based PSCs afford a lower apparent trap density of states (tDOS) at about 0.4 eV (Fig. 10d), which could be attributed to the lesser number of ionic charges or the reduction of electronic defects in perovskite.
Introduction of MACl as an additive can afford void-free perovskite-buried interface for scalable processing of perovskite solar modules. Hu et al.160 have demonstrated a void-free perovskite-buried interface for scalable processing of p-i-n-based perovskite solar modules (>10 cm2) with FAPbI3 as perovskite. The growth of the perovskite film is controlled by utilizing a moderate flow of N2 during vacuum-assisted growth, while the utilization of MACl as an additive hinders interfacial voids in blade-coated large-area perovskites. As a result, the PSC exhibits a high PCE of 22.3%, while it exhibits high stability, while the scalable perovskite solar module with 12.25 cm2 of aperture area exhibits a high PCE of 18.3% with a geometric fill factor of 96.3%.
The utilization of interfacial ligand confinement strategy can provide top-contact passivation and improve the extraction of electrons at the electron selective contact, and that can enhance the performance of the PSCs. Huang etal.161 have demonstrated the fabrication of finite 2D/3D hierarchical perovskite structures through interfacial ligand confinement to enhance the stability and efficiency of the inverted PSCs and modules. The generation of a finite and dense 2D layer is achieved due to the utilization of a diamine masking reagent atop the 3D perovskite layer, which can modulate cation exchange through hydrogen bonding, while this can provide top-contact passivation and improve the extraction of electrons at the electron selective contact. Hence, the PSC exhibits a remarkable PCE of 24.7% with high stability (>90% retention for 1000 h) according to the MPP tracking test.
The utilization of suitable amphoteric organic salt can modulate the energy level alignment and film morphology at the perovskite buried interface. Zhang et al.162 have utilized 2-(4-fluorophenyl)ethylammonium-4-methyl benzenesulfonate (4FPEAPSA) to modulate the energy level alignment and film morphology at the perovskite buried interface. The amphoteric organic 4FPEAPSA salt treatment enhances the growth of a coarse-grained, void-free, and hydrophobic film by inducing crystal orientation. Thus, the 4FPEAPSA with dual functionality can interact chemically with the perovskite film to passivate the formamidine vacancies and defects of iodine, returning the fermi level of perovskite to its defect-free state. Moreover, the interfacial charge extraction and transport of PSC can be enhanced by the generation of a p-type doping buried interface to attain diminished carrier recombination loss. This PSC exhibits a large PCE of 25.03% while it exhibits high stability.
Utilization of conductive and transparent oxide perovskite as ETL can afford high lattice matching, and obviate the generation of a deteriorated buried interface, which can provide less strain, less defects, high crystallinity, fewer voids, and suppress ion migration, and that can enhance the performance of the PSCs. Luo et al.163 have utilized the SrSnO3 perovskite as ETL rather than using a passivator, where the SrSnO3 is a conductive and transparent oxide perovskite. Thus, a well-ordered beginning of the growth of perovskite on the ETL is achieved due to the high lattice matching, hindering the generation of a deteriorated buried interface. Hence, the buried interface affords reduced strain, less defects, high crystallinity, fewer voids, and suppressed ion migration. Consequently, the PSC exhibits an excellent PCE of 25.17%, and retain high stability (90% retention for 1000 h) according to the MPP tracking test.
Utilization of suitable surfactant on the surface of the TiO2 can diminish surface energy, enhance the carrier-extraction efficiency, relieve the compressive stresses at the buried interface, and that can enhance the performance of the PSCs. Su et al.164 have utilized heptadecafluorooctanesulfonate tetraethylammonium (HFSTA) as a surfactant on the surface of the TiO2. The DFT calculations reveal that the generation of O vacancies is inhibited on HFSTA-TiO2, and that can diminish surface energy and enhance the carrier-extraction efficiency. The experimental studies disclose that the enhanced crystallization process and diminished heterogeneous nucleation sites enable the generation of good-quality perovskite films, while the compressive stresses at the buried interface are significantly relieved by the flexible long alkyl chains in HFSTA. Thus, the PSC exhibits a high PCE of 25.03% due to the passivation of TiO2, stress relief, and modulation in the crystallization process, while it exhibits high stability (66% retention for 50 h) by MPP tracking test.
Utilization of a pre-embedding mixed A-cation halide strategy can enable the formation of the high-stable 3D perovskite from the residual unstable PbI2 near the buried interface, optimize the lattice strain of the perovskite near the buried interface, inhibit the detrimental δ phase formation from the α phase, and enhance the phase stability of the perovskite. Gao et al.165 have improved the stability of the PSC through utilizing a pre-embedding mixed A-cation halide strategy, where the high-stable 3D perovskite is formed from the residual unstable PbI2 near the buried interface. Thus, this strategy could optimize the lattice strain of the perovskite near the buried interface, inhibit the detrimental δ phase formation from the α phase, and enhance the phase stability of the perovskite. As a result, the PSC exhibits a high PCE of 24.26%, while it exhibits high stability (94.7% retention for 1000 h) by MPP tracking test.
Strategies associated with buried interfaces including the utilization of suitable additives64,160, amphiphilic molecular hole transporter81, amphoteric organic salt162, conductive and transparent oxide perovskite as ETL163, suitable surfactant on the surface of the TiO2164, interfacial ligand confinement161, and a pre-embedding mixed A-cation halide strategy165, enhanced the efficiency and/or stability of the PSCs.
Strategies for ion migration for PSCs
Various strategies have been performed to retard the ion migration to enhance the performance of PSCs. Utilization of suitable material as iodine trapper can alleviate the ion migration, and that can enhance the stability of PSCs. Li et al.70 have utilized β-cyclodextrin as an iodine trapper to enhance the stability of PSCs. The iodine formed in perovskite can be trapped by β-cyclodextrin, and the bowl structure of β-cyclodextrin hinders the escape of iodine, where the trapped I2 can react with and consume Pb0 defects in perovskite films, further enhancing the stability of PSCs. Therefore, the PSC exhibits a high PCE of 23.2%, while it exhibits high stability (∼90% retention for 1000 h (Table 9) under ISOSL-1 by MPP tracking test at 25 °C; ∼81.2% retention for 300 h under ISOSL-2 by MPP tracking test at 85 °C, where ISOS represents International summit on organic photovoltaic stability).
Substitution of lead with tin in perovskite can suppress the ionic transport, and that can enhance the performance of the PSCs. Dey et al.166 have demonstrated that ionic transport is suppressed in halide perovskite optoelectronics due to the substitution of lead with tin. The scan-rate-dependent current-voltage investigations on mixed Pb-Sn and Pb PSCs disclose losses for short-circuit current at lesser scan rates in both kinds of perovskites, which can be traced to the existence of mobile ions. Temperature-dependent impedance spectroscopy measurements and scan-rate-dependent hysteresis analyses reveal that the ion migration in Pb-Sn PSCs is suppressed when compared to that of Pb perovskite solar cells. As shown in Fig. 11a–c, three distinct pathways for iodide ion migration are examined, which are labeled as A, B, and C, while the Fig. 11d depicts their corresponding energy profiles. The A, B, and C pathways for FAPb0.5Sn0.5I3 and FASnI3 exhibit various orders of equatorial-axial type hops, where >0.9 eV of energy barriers for both perovskites and 1.45 eV for FAPb0.5Sn0.5I3 and 1.12 eV for FASnI3 of rate-limiting ion migration energies for long-range diffusion are obtained. Thus, the simulations obviously suggest that the iodide migration adjacent to Sn vacancy defects in FASnI3 and FAPb0.5Sn0.5I3 exhibits higher energy barriers of >1.1 eV when compared to that of Pb perovskites with <0.5 eV because of the severe local structural distortion of Sn vacancies, thereby diminishing halide ion mobility. The theoretical calculations and experimental investigations disclose that the Sn vacancies with local structural distortions in mixed Pb-Sn perovskite play a vital role in increasing the suppression of iodide ion migration.
a–c Inequivalent iodide migration paths in FAPb0.5Sn0.5I3, which involve the SnI2 Schottky-type defect composed of two I vacancies and a Sn vacancy (the colors of spheres represent yellow for lead, olive for tin, pink for iodine, turquoise for carbon, blue for nitrogen, and white for hydrogen atoms, where the direction of iodide migration in the lattice is indicated by the red arrows); d Energy profiles for iodide ion migration through different paths (a–c) in FAPb0.5Sn0.5I3 (reproduced with permission from ref. 166 Copyright 2024, The Royal Society of Chemistry).
Utilization of a supermolecule as a buffer layer at the interface of perovskite can enhance defect self-healing and inhibit ion migration, and that can enhance the performance of the PSCs. Zhang et al.167 have utilized a supermolecule (starch-polyiodide) as a buffer layer at the interface of perovskite, and this bifunctional buffer layer can enhance defect self-healing and inhibit ion migration. Hence, the metastable dynamics of degradation in perovskites can be tuned by the supramolecular chemistry. Therefore, the PSC exhibits a high PCE of 24.3%, and it exhibits high stability (92% retention for 1472 h by MPP tracking test; 98% retention for 42 diurnal cycles (12 h light/12 h dark cycle), while it exhibits intense electroluminescence with high external quantum efficiencies (>12%).
Introduction of suitable alkali metals can effectively control the ionic migration due to an electrostatic interaction with the organic compounds, and that can enhance the performance of the PSCs. Aranda et al.168 have demonstrated the control of ionic migration in PSCs by alkali metals. Thus, the role of Na, Li, and K on the photovoltage of the MAPbBr3 (wide band-gap perovskite) is systematically examined by utilizing negative capacitance in impedance spectroscopy as a tool. Sodium cations inhibit adverse interfacial recombination pathways, and it exhibits a higher stabilized VOC of 1.65 V when compared to that of Li and K. Impedance studies disclose that the sodium substantially affects the material’s bulk, which is corroborated with the XPS and time-of-flight secondary ion mass spectrometry studies. These investigations reveal that the ionic migration is effectively diminished by Na in perovskite materials. The XPS studies disclose that Na effectively decreases the ionic migration in perovskite materials due to an electrostatic interaction with the organic compounds.
Utilization of bis-diazirine molecules can suppress the ionic migration due to the covalent bond formation with the organic cations of perovskites, and that can enhance the performance of the PSCs. Liu et al.169 have utilized bis-diazirine molecules, where the organic cations of perovskites can be covalently bonded with bis-diazirine molecules to alleviate the escape of organic components from perovskites, and that can hinder adverse effects including more defects, organic vapors, and ion migration. Ab initio simulation and experimental investigations disclose that the bis-diazirine molecules effectively immobilize the organic cations, leading to an improvement in the illumination, thermal, and electrical bias resistance properties of perovskites. Hence, the PSC exhibits a large PCE of 23.95%, while it exhibits high stability (98.6% retention for 1000 h by MPP tracking test).
Introduction of a conformally bonded molecular interface can mitigate iodine migration and enhance the performance of PSCs. Yuan et al.170 have utilized a conformally bonded molecular interface to mitigate iodine migration and enhance the performance of PSCs. Thus, three linear molecules (L1, L2, and L3) with a D-A-D’-A-D structure as dopant-free HTMs are obtained by enlarging the indacenodithiophene π-bridge donor (D’) to merge with the benzothiadiazole acceptor (A) and methoxy triphenylamine donor (D). The intermolecular packing effect and intramolecular dipole effect are leveraged by the π-bridge extension with active sites for coordination, leading to a conformally bonded ultrathin interface with uniform and compact coverage (about 60 nm) to guard the buried perovskite and hinder iodine migration. Ultrafast laser spectroscopy investigations disclose that L-series molecules possess a sequential decline of photoexcited energy disorder to demonstrate the structure-performance-stability relationship. Hence, the n-i-p PSC with L3 (ITO/SnO2/perovskite/L3/MoO3/Au) exhibits a high PCE of 22.61%, while it exhibits high stability (>85% retention for 500 h at 85 °C by MPP tracking test).
Incorporating a cross-linked polymer between 2D and 3D perovskite can reduce the ionic diffusion, and that can enhance the performance of the PSCs. Luo et al.82 have designed a 3D perovskite/cross-linked polymer/2D perovskite heterostructure to reduce the ionic diffusion between the surface 2D and bulk 3D perovskites by adding a cross-linked polymer to the top of a 3D perovskite layer and then depositing a 2D perovskite layer using a vapor-assisted two-step process. The heterostructure is sustained by the cross-linked polymer by delaying the diffusion of FA+ and 4F-PEA+ cations across the 2D and 3D perovskites, according to thickness-profiled elemental analysis and photoluminescence spectra (FA+: formamidinium; 4F-PEA+: 4-fluorophenylethylammonium). Consequently, the PSC has a high PCE of 21.2% and great stability (retention of 90% for 4390 h at 60 °C according to the MPP tracking test).
Strategies to retard ion migration, including the utilization of β-cyclodextrin as an iodine trapper70, a buffer layer (a starch-polyiodide supermolecule) at the perovskite interface167, alkali metals168, bis-diazirine molecules169, a conformally bonded molecular interface170, incorporating a cross-linked polymer between 2D and 3D perovskite82, and substitution of lead with tin in perovskite166 enhanced the efficiency and/or stability of the PSCs.
HTL and ETL-related strategies for PSCs
Since the design of (opto)electronic devices resistant to heat is highly desirable, the formation of stable and compact semiconducting thin films is required. Nevertheless, developing solution-processable organic semiconductors with superior thermo-mechanical properties is a highly challenging task. Fang et al.65 have demonstrated that the organic semiconductors (obtained through solution-processable route) exhibit significantly high glass transition temperatures (>220 °C), which could be due to the selection of tribenzotriazatruxene with helical isomer instead of planar isomer as the conjugated core. Microscopic studies disclose that the integration of a non-planar helicene as the conjugated core with organic semiconductors can be solution processed into integral thin films and morphologically homogenous on several substrates, due to a remarkably enhanced solubility of >200 mg mL−1. Furthermore, the theoretical and experimental studies disclose that the helical tribenzotriazatruxene-based organic semiconductor exhibits considerably high water resistance and hole conduction, possibly due to the heightened molecular packing density. As a result, the n-i-p type PSC exhibits a high PCE of 23.9% (Table 10), and high stability (93% retention for 500 h) by MPP tracking test, and high long-term thermal stability at 85 °C.
Utilization of suitable spirobifluorene-based hole transport material containing an electron-donor can afford high glass transition temperature, and provide comparable HOMO energy level, and that can enhance the thermal stability and performance of the PSCs. Ren et al.171 have fabricated a spirobifluorene-based hole transport material containing an electron-donor of extremely asymmetric fluorenylcarbazolamine (SBF-FC) to achieve high thermal stability and efficiency in PSC. Thus, the SBF-FC exhibits a very high glass transition temperature, while it exhibits a comparable HOMO energy level when compared to that of spiro-OMeTAD. A composite is prepared by blending an 85:15 weight ratio of SBF-FC and 4-tertbutylpyridinium bis(trifluoromethanesulfonyl)imide, while the composite exhibits a high conductivity of 49 mS cm−1 at room temperature and retains a high glass transition temperature of 176 °C. Furthermore, the PSC with SBF-FC based hole transport layer on a FAPbI3 thin perovskite film affords highly homogenous morphology and high thermal stability, and it effectively inhibits the decomposition and corrosion of the perovskite film. As a result, the PSC exhibits a high PCE of 24.7%, high stability (89% retention for 200 h) by MPP tracking test, and high long-term thermal stability at 85 °C.
Utilizing suitable polymeric hole transport material can afford superior hole-extraction capacity, reduce the interfacial recombination, stabilize the interface between perovskite and hole transport material, and that can enhance the performance of the PSCs. Ren et al.172 have demonstrated that the polymerizing of carbazole phosphonic acid (PACz) into Poly-4PACz is an efficient polymeric hole transport material (HTM), where it affords superior hole-extraction capacity and can further reduce the interfacial recombination and stabilize the interface between perovskite and hole transport material. Furthermore, Poly-4PACz is insensitive to layer thickness on both fluorine-doped tin oxide and indium-doped tin oxide substrates, while still affording high conductance. These are promising characteristics for the scalable fabrication of perovskite films. As a result, the blade-coated p-i-n type perovskite solar cell and module exhibit high PCE of 24.4% (6.84 mm2) and 20.7% (25.0 cm2), respectively, while the PSC exhibits high stability (94% retention for 1500 h) by MPP tracking test.
Printable planar carbon electrodes are considered a promising rear contact for perovskite solar cells when compared to metal electrodes prepared by thermal evaporation, whereas PSCs with carbon-electrode exhibit fewer PCEs than PSCs with metal-electrode. Utilizing a hole-transporting bilayer strategy can diminish the surface recombination of the perovskite, enhance the hole extraction, and that can improve the performance of the PSC with carbon-electrode. Du et al.173 have simultaneously improved the open-circuit voltage and fill factor of PSC with carbon-electrode using a hole-transporting bilayer (HTbL) strategy. The HTbL has been fabricated by the blade coating of an organic semiconductor followed by another organic semiconductor between perovskite and carbon, where the surface recombination of the perovskite is diminished by the inner hole transport layer (HTL), while the hole extraction to carbon is improved by the outer HTL. The PSC with carbon-electrode exhibits impressive PCE of 19.2% and high stability (negligible decay for 2500 h) according to the MPP tracking test.
Introduction of lead chelation molecules into HTLs can reduce the amorphous area in perovskites adjacent to HTLs and passivate the bottom perovskite surface, and that can enhance the performance of the PSC. Fei et al.174 have observed a diminished amorphous area in perovskites adjacent to HTLs and a passivated bottom perovskite surface after the introduction of lead chelation molecules into HTLs due to the strong interaction with Pb2+. Therefore, the PSC and module exhibit unexpected PCE of 24.6% and 21.8% (26.9 cm2) with high stability (>90% retention for 1500 h) by MPP tracking test, and it shows high light-soaking stability for 3010 h.
Utilization of dual functional ionic liquids as a dopant and surface passivator for HTL can enhance charge extraction and diminish non-radiative recombination, and that can enhance the performance of the PSC. Kim et al.175 have utilized dual functional ionic liquids (ILs) as a dopant and surface passivator for hole transporting layer (HTL) to attain efficient and stable PSCs and modules. The dual functional ionic liquid dopants is prepared through a simple acid–base reaction between alkylamines and bis(trifluoromethane)sulfonimide (HTFSI), where the alkylamines are butylamine, hexylamine, octylamine, and decylamine, while the dual functional ionic liquid dopants (alkylammonium bis(trifluoromethylsulfonyl)imides or alkylammonium TFSIs) are BATFSI, HATFSI, OATFSI, and DATFSI. The OATFSI affords adequate miscibility with a poly(triarylamine) solution among several ILs, which causes a smoother morphology of the HTL with improved electrical property through efficient doping. Furthermore, the in situ passivation of the perovskite surface by OATFSI occurs during the spin-coating deposition of the hole transport layer. As a result of enhanced charge extraction and diminished non-radiative recombination, the OATFSI based PSC with a mesoporous n-i-p structure exhibits a high PCE of 23.34%, and the perovskite solar modules exhibit high PCE of 19.91% (209.39 cm2) and 18.54% (224.89 cm2), while the PSC exhibits high stability (~89% retention for 1080 h) by MPP tracking test.
Self-assembling of Me-4PACz onto NiOx nanoparticles (Me-4PACz: 4-(3,6-dimethyl-9H-carbazol-9-yl)butyl]phosphonic acid) can generate an efficient HTL, where the homogenized NiOx nanoparticles can generate Ni3+ and surface hydroxyl groups for bonding, and afford high conductivity, and that can enhance the performance of the PSC. Yu et al.176 have demonstrated an efficient HTL by self-assembly of Me-4PACz onto NiOx nanoparticles via hydrogen peroxide treatment. Thus, a well uniform dispersion of nanoparticles with high conductivity is achieved, possibly due to the generation of Ni3+ and surface hydroxyl groups for bonding. As a result, the perovskite solar cell and mini-module attain high PCE of 25.2% (0.074 cm2) and 21.0% (14.65 cm2), respectively, while the PSC exhibits high stability (85.4% retention for 1000 h at ∼50 °C by MPP tracking test; 85.1% retention for 500 h of accelerated aging at 85 °C).
Utilization of co-adsorbent strategy can disassemble high-order clusters, which can uniformly distribute the phosphonic acid molecules, and that can enhance electronic structures and diminish interfacial recombination, and that can enhance the performance of the PSCs. Park et al.177 have demonstrated hole-selective contact of a conformal self-assembled monolayer (SAM) on light-managing textured substrates. The molecular dynamics simulations disclose that incomplete SAM coverage can occur due to cluster generation during the adsorption of phosphonic acid. Therefore, a co-adsorbent strategy is devised to disassemble high-order clusters, leading to a uniform distribution of phosphonic acid molecules, which can enhance electronic structures and diminish interfacial recombination. Hence, the PCE for the PSC reaches as high as 24.8% (Shockley-Queisser maximum: 95%), while it exhibits high stability (95% retention for 1075 h) by MPP tracking test. The reason for the interaction of phosphonic acids with textured transparent conductive oxides (TCOs) is deeply investigated. Therefore, the interaction of 2PACz (Fig. 12a) with fluorine-doped tin oxides (FTO) is investigated (2PACz: 2-(9H-carbazol-9-yl)ethyl)phosphonic acid). 2PACz is widely used as a hole-selective contact in PSCs, possibly due to its much highest occupied molecular orbital level and superior defect passivation. Using SnO2, the surface properties of fluorine-doped tin oxides are approximated. Corrugated versus flat SnO2 is investigated to determine the effects of surface morphology. As shown in Fig. 12b, the molecular dynamics simulations disclose that agglomeration can occur in 2PACz molecules, resulting in the formation of dimers, trimers, and tetramers. As shown in Fig. 12b, 90% of the final number of clusters is attained within the initial 1 nanosecond of simulations, where the dimmers are the most common among those clusters, as depicted in Fig. 12c. The textured surfaces exhibit more clusters when compared to flat surfaces. The formation of clusters is closely associated with the inhomogeneity of SAMs on FTO. As shown in Fig. 12d, on top of textured SnO2 surfaces, 162 2PACz molecules have been positioned, while clear phase segregation is observed on the final equilibrated structures, whereas ∼85% of the SnO2 surface is not wrapped with any 2PACz molecules. This case is also associated with a large system using 1134 2PACz molecules (22 nm width). The 3-mercaptopropionic acid (3-MPA; Fig. 12a) is introduced in simulations to break apart 2PACz clusters, especially on textured substrates, because a –SH (thiol group) could interact with a carboxyl group and phosphonic acids attached to FTO. Thus, a co-adsorbent strategy is applied to disassemble high-order clusters. Therefore, a slower formation rate of 20 ns−1 versus 28 ns−1 and a comparatively 15% diminish in the number of clusters on textured surfaces (Fig. 12b) are achieved after mixing 3-MPA with 2PACz at a molar ratio of 1:9. Especially the higher-order clusters such as trimers and tetramers are diminished to15.3%, as shown in Fig. 12c. Thus, the extended surface coverage of 67% versus 15% is achieved by 2PACz molecules due to the formation of less phase segregation in the mixed system. The ab initio molecular dynamics (AIMD) simulations disclose that the free localization of individual 2PACz molecules is inhibited after the introduction of 3-MPA due to the formation of a supramolecular structure, as depicted in Fig. 12f. Therefore, the agglomeration between as-formed dimmers is diminished, leading to the generation of higher-order clusters. The DFT calculations disclosed that the 2PACz and 3-MPA exhibit surface binding energies of −3.2 eV and −2.6 eV, respectively, which are much larger than the interaction energies of 2PACz clusters (−0.3 to −0.1 eV per molecule). This represents strong anchoring of 3-MPA and 2PACz molecules on contact with substrate surfaces.
a Chemical structures of the bifunctional compound 3-MPA and the phosphonic acid 2PACz; b Total number of 2PACz clusters generated for 10 ns with (control) and without (mixed) 3-MPA; c Varieties of 2PACz clusters generated at equilibrium; Equilibrated molecular representations (Top views) of d control and e mixed systems (2PACz: Pink; 3-MPA: blue; Sn atoms in the background: Yellow; O atoms in the background: Red); f Successive steps with an AIMD trajectory depicting the role of a co-adsorbent (3-MPA), big, encircled regions are magnifications of small encircled areas (reproduced with permission from ref. 177 Copyright 2023, Springer Nature).
Utilization of suitable self-assembled monolayer based HTL can enhance the ion-blocking properties, which can enhance the operational stability of PSCs at elevated temperatures. Jiang et al.178 have demonstrated that their six-month outdoor aging tests can be predicted by indoor accelerated stability tests using an inverted PSC stack holding a PCE of 25.58%. The degradation rates of PSCs at elevated temperatures and under illumination are very helpful in determining the reliability of outdoor devices. Furthermore, the operation stability of the PSCs is strongly affected by the ITO/self-assembled monolayer-based HTL/perovskite interface (ITO: indium tin oxide). The averaged operational stability of PSC from 50 °C to 85 °C is increased by a factor of about 2.8 by enhancing the ion-blocking properties of the self-assembled monolayer-based HTL, leading to a projected 20% degradation at 85 °C for >1000 h and at 50 °C for ∼8200 h.
The effective hole-selective layer (HSL) with high thermal stability has been demonstrated by Li et al.83. It is made of a film of NiOx nanoparticles combined with a surface-anchored MeO-4PADBC SAM (MeO-4PADBC: 4-(3,11-dimethoxy-7H-dibenzo[c,g]carbazol-7-yl)butyl)phosphonic acid) and has the ability to stabilize and improve the NiOx/perovskite interface. The voltage deficit of perovskite solar cells with different perovskite compositions was reduced by the good contact, binding, and energetic alignment between perovskite and NiOx/MeO-4PADBC, which results in strong interface toughening effects during heat stress. Consequently, the 1.53 eV PSC shows a high PCE of 25.6% and high stability (>90% retention for 1200 h) according to the MPP tracking test. The interfacial interactions between SAMs and perovskite can be modulated by tuning the terminal functional group. Thus, the efficiency of the PSC can be enhanced in some cases due to the enhancement in interfacial contact after the introduction of two methoxy groups (OMe), whereas the introduction of OMe can also decrease the dipole moment in some other cases. Due to the substitution of two methoxy groups with opposing directions of dipole moment, the MeO-2PACz exhibits a net molecular dipole moment near to zero, as illustrated in Fig. 13a. This behavior can be attributed to the high symmetry and planarity of the carbazole structure. Therefore, a new SAM of MeO-4PADBC, as depicted in Fig. 13b is designed by utilizing a non-coplanar screw-shaped DBC unit as the core (DBC: Dibenzo[c,g]carbazole) to alleviate the adverse outcome on the dipole moment after substituting methoxy groups. The DFT calculations disclose that the MeO-4PADBC exhibits a dipole moment of 2.4 D, which is slightly less than that of 2.9 D for 4PADBC (4PADBC: 4-(7H-dibenzo[c,g]carbazol-7-yl)butyl)phosphonic acid), which is entirely different from those observed for carbazole-based SAMs. As depicted in Fig. 13c, the NiOx/MeO-4PADBC SAM as HSL is compared with the MeO-4PADBC SAM for inverted PSCs. In the FTIR spectra (Fig. 13d), the absorption peaks for P-OH (1035 and 950 cm−1) and P = O (1169 cm−1) of NiOx/MeO-4PADBC show a noticeable shift in comparison with MeO-4PADBC, suggesting the generation of a chemical bond. The existence of the chemical bonding is further confirmed by XPS spectra, where a peak shift is observed at the Ni 2p core level of NiOx/MeO-4PADBC and NiOx films deposited on ITO substrates. The Cs0.05FA0.85MA0.1PbI3 perovskite film exhibits almost similar crystal structure and absorbance on both substrates, whereas larger crystal domains are observed for the perovskite on the ITO/NiOx/MeO-4PADBC substrate when compared to that of the control substrates. As a result, SAM molecules densely anchor on the ITO/NiOx substrate by stronger tridentate binding absorption. This can reduce the substrate’s surface roughness, offer a more hydrophobic surface, and strengthen the bond between SAM molecules and perovskite. Hence, the perovskite crystal nucleation and growth are enhanced, and that improves the perovskite crystallization. In the UPS spectra (UPS: Ultraviolet photoelectron spectroscopy), the energetic alignment of different substrates with respect to perovskite absorbers with different bandgaps (1.8, 1.68, and 1.53 eV) is shown in Fig. 13e, while the corresponding results are provided in a schematic diagram (Fig. 13F). The ITO/NiOx/MeO-4PADBC and ITO/MeO-4PADBC exhibit the HOMO energy levels of −5.45 eV and −5.34 eV, respectively, and the ITO/NiOx/MeO-4PADBC exhibits a deeper work function (Φ) of −4.95 eV when compared to that of the ITO/MeO-4PADBC (−4.90 eV), suggesting more suitable energetic alignment with various perovskite absorbers. Nevertheless, in the case of carbazole-based SAM molecules onto the ITO/NiOx surface, the work function Φ is shifted upward for the ITO/NiOx/MeO-2PACz system, which is in contrast to the ITO/NiOx/2PACz substrate. This phenomenon can be ascribed to the significantly reduced dipole moment after the substitution of OMe groups on 2PACz, where this reduction can be attributed to the highly planar carbazole motif, as mentioned earlier. Time-resolved photoluminescence (TRPL) decay data disclosed that perovskite on ITO/NiOx/MeO-4PADBC exhibits the lowest carrier lifetime of 32.3 ns in comparison with perovskite on ITO/MeO-4PADBC (42.9 ns) and ITO (925.5 ns), suggesting enhanced hole extraction with the ITO/NiOx/MeO-4PADBC substrate.
a Molecular structure and side view of MeO-2PACz; b Molecular structure and side view of MeO-4PADBC; c Schematic diagram illustrates the anchoring of MeO-4PADBC on NiOx nanoparticle as the HSL in PSC; d FTIR spectrum of NiOx/MeO-4PADBC in comparison with MeO-4PADBC. e UPS spectrum of ITO/NiOx/MeO-4PADBC in comparison with ITO/NiOx and ITO/MeO-4PADBC, where the right side shows the UPS spectra in the VB region (VB: valence band), and the left side shows the UPS spectra around the secondary electron cutoff (WF work function); f Schematic illustration of the band edge positions of the HSLs based on UPS spectra, referenced to the vacuum level (EF Fermi level, EVAC Vacuum level, EVBM energy of valence band maximum, ECBM energy of conduction band minimum) (reproduced with permission from ref. 83 Copyright 2023, American Association for the Advancement of Science).
Utilization of suitable self-assembled cocrystal layer can diminish interfacial charge accumulation and optimize interfacial energy level alignment through a strong charge-transfer effect, and that can enhance the performance of the PSCs. Wang et al.179 have fabricated a self-assembled cocrystal layer (SAM-CL) to enhance the performance of PSCs, where the SAM-CL is obtained from the PRMA monolayer on the perovskite surface and F4TCNQ doped in spiro-OMeTAD by intermolecular π–π interactions and hydrogen bonds (PRMA: 1-pyrenemethylamine hydrochloride; F4TCNQ: 2,3,5,6-tetrafluoro-7,70,8,80-tetracyanoquinodimethane). Thus, interfacial charge accumulation is reduced due to the optimization of interfacial energy level alignment by SAM-CL through a strong charge-transfer effect. Furthermore, moisture invasion and ion migration are effectively inhibited by the superior barrier effect of F atoms and huge pyrene rings in SAM-CL, thereby enhancing the stability of PSCs. As a result, the PSC exhibits a high PCE of 24.03% while it exhibits high stability.
Introduction of suitable interlayer between perovskite and HTL can alleviate redox reactions at the interlayer/perovskite interface, inhibit non-radiative recombination in the perovskite layer, enable efficient charge extraction, improve the depletion at the interface of perovskite and HTL, and that can enhance the performance of the PSCs. Li et al.180 have designed an interlayer between perovskite and HTL to enhance the efficiency and stability of inverted PSCs, where the interlayer is composed of ATOx combined with a SAM (ATOx: p-type antimony-doped tin oxides). Thus, the chemical stability of the interface is improved by ATOx, while negligible redox reactions are observed at the ATOx/perovskite interface when compared to those at the NiOx/perovskite interface. Furthermore, non-radiative recombination is inhibited by ATOx in the perovskite layer, and thus efficient charge extraction is achieved by improving the depletion at the interface of perovskite and HTL. As a result, these enhancements lead to a PSC that not only achieves an impressive power conversion efficiency (PCE) of 25.7% but also maintains high stability, retaining 93% of its initial performance after 500 h as demonstrated in the MPP tracking tests.
Developing a strategy for the co-deposition of the hole-selective contact and absorber can simplify the manufacturability towards the commercialization of efficient PSCs. The co-deposition can be enabled by the introduction of carboxylic or phosphonic acids into perovskite precursor solutions that can self-assemble on the ITO substrate during the processing of perovskite film. Thus, a strong self-assembled monolayer of superior HSC can be formed while the perovskite can be crystallized. Zheng et al.181 have designed a facile one-step solution-coating procedure to co-deposit the hole-selective contact (HSC) and absorber towards the commercialization of efficient inverted PSCs, where HSC and perovskite light absorbers are spontaneously obtained. The introduction of carboxylic or phosphonic acids into perovskite precursor solutions self-assembles on the ITO substrate during the processing of perovskite film. Thus, a strong self-assembled monolayer of superior HSC is formed while the perovskite is crystallized. This strategy obviates wettability issues, and thus facile device fabrication is achieved, thereby simplifying the manufacturability of PSCs. This strategy exhibits good generality, where it is compatible with various perovskites, SAM molecular systems, solvents, and processing methods. Therefore, the PSC exhibits a high PCE of 24.5% and stability (>90% retention for 1200 h under continuous illumination) according to the MPP tracking test.
Utilization of suitable hole-shuttle bifunctional molecule can enable favorable energy level alignment with the perovskite, diminish the voltage losses, passivate the defects at the interface of perovskite/HTL, and that can enhance the performance of the PSCs. You et al.182 have demonstrated that the bifunctional molecule CBz-PAI acts as a hole-shuttle molecule for passivate the defects at the interface of perovskite/HTL, where the CBz-PAI is composed of phenylammonium iodide and carbazole-triphenylamine units (CBz-PAI: 4-(3,6-bis(4-(bis(4-methoxyphenyl)amino)phenyl)-9H-carbazol-9-yl)benzenaminium iodide). The CBz-PAI plays as a hole shuttle between the HTL and the perovskite layer due to a favorable energy level alignment with the perovskite. Thus, the external device Voc and the difference between the quasi-Fermi level splitting of the perovskite, or ‘internal’ Voc, are diminished, and that diminishes the voltage losses. Hence, the PSC exhibits a large PCE of 24.7%, while it exhibits high stability (94.6% retention for 1100 h by MPP tracking test; 92.3% retention for 1000 h at 85 °C under damp heat test in 85% relative humidity).
Besides, tuning the ETL can modulate the performance of the PSCs. The combination of compact-TiO2 with mesoscopic-TiO2 and SAM can enhance the photocarrier extraction at the interface of ETL/perovskite, and that can enhance the performance of the PSCs. Yang et al.183 have investigated the combination effects of c-TiO2 as ETL (c-TiO2: compact TiO2) with and without mesoscopic TiO2 (m-TiO2) on top, and iodine-terminated silane SAM on the photovoltaic performance, operational stability, mechanical behavior, and opto-electronic properties of metal-halide perovskites-based PSCs. Thus, c-TiO2 combined with m-TiO2 and SAM presents an almost threefold increase in interfacial toughness when compared to that of c-TiO2. This is ascribed to the synergistic effect of the m-TiO2/perovskite nanocomposite at the interface and the improved adhesion provided by the iodine-terminated silane SAM. Thus, the combination of c-TiO2 with m-TiO2 and SAM exhibits better photocarrier extraction at the interface of ETL/perovskite, and thus the PSC exhibits a high PCE of 24.12% while it exhibits high stability (~90% retention for ~1200 h by MPP tracking test; extrapolated T80 stability: 80% retention for ≈18,000 h).
Light soaking can obviously influence the efficiency and stability of the PSCs. In large-area inverted PSCs, the light-soaking is obviously diminished when C60 is used rather than PC61BM as ETL, where a thin BCP interlayer is used between ETL and contact electrode (BCP: bathocuproine). Henderson et al.66 have demonstrated the vital molecular origins of the light-soaking effect through a combination of ambient photoemission spectroscopy, surface photovoltage, and Raman spectroscopy, integrated with DFT simulations. A photoinduced charge-transfer complex is formed by BCP with both PC61BM and C60. The C60 dimer formation is accelerated by the C60/BCP complex, which affords better electron extraction and diminishes recombination loss. However, the PC61BM dimer formation is inhibited by the PC61BM/BCP complex, suggesting that the dimerization of PC61BM is not due to light-soaking. Besides, it is the slow light-stimulated generation of the PC61BM/BCP charge-transfer complex itself and the new energetic transport levels related to it that lead to the much slower and robust light-soaking effect of PC61BM based perovskite solar cells.
Several HTL-related strategies including polymeric hole transport material172, introduction of lead chelation molecules into HTLs174, dual functional ionic liquids as a dopant and surface passivator for HTL175, homogenized NiOx nanoparticles176, NiOx nanoparticle film with a surface-anchored suitable SAM83, self-assembled cocrystal layer179, suitable interlayer between perovskite and HTL180, co-deposition of the hole-selective contact and absorber181, a hole-shuttle bifunctional molecule for defects passivation at the interface of perovskite/HTL182, hole-transporting bilayer strategy173, and co-adsorbent strategy177 enhanced the efficiency and/or stability of the PSCs. Tuning of ETL66,183 enhanced the efficiency and/or stability of the PSCs.
Interlayer/molecular bridge/buried-metal-grid electrode strategies for PSCs
The design of suitable interlayer, molecular bridge, or buried-metal-grid electrodes can elevate the performance of PSCs. Utilization of suitable multifunctional and chemically stable buffer material between ETL and metal electrode can enable sufficient electrons flow from the ETL to the electrode, alleviate the inter-diffusion of harmful species into the perovskite, prevent the degradation products out of the perovskite, and that can enhance the performance of the PSCs. Chen et al.73 have utilized amorphous ytterbium oxide (YbOx (thickness: ∼1.5 nm); Fig. 14b) as a multifunctional and chemically stable buffer material between ETL and metal electrodes for inverted PSCs through scalable thermal evaporation deposition. The schematic illustration (Fig. 14a) of the charge-carrier transport across the electron-selective layer/YbOx/Cu interface shows that when electrons flow to the Cu electrode from the ETL (C60), phonon-assisted electron hopping occurs due to vacant localized states of the buffer layer (YbOx). The carrier transport in the buffer layer (YbOx) is caused by thermally activated hopping to another state from one localized state. As a result of YbOx buffer, the inverted PSCs with a narrow-bandgap (NBG), wide-bandgap (WBG), and mid-bandgap (MBG) perovskite absorber exhibit high PCE of 25.2% (Fig. 14c; Table 11), 20.1% (Fig. 14e), and 22.1% (Fig. 14d), respectively, with high reproducibility (Fig. 14f), while it exhibits high stability (97% retention for 1000 h by MPP tracking test; 85% retention for 500 h at 85 °C in ambient air with 50% relative humidity by MPP tracking test (ISOS-L-3 accelerated aging)). The PSCs with YbOx buffer exhibit a very high VOC (Fig. 14g), which is almost close to or even exceeds 90% of the radiative limit versus the bandgap energy. Additionally, the external quantum efficiencies of the electroluminescence (EQEELs) for the PSCs with YbOx buffer are determined, where the PSC with NBG perovskite absorber exhibits 5.12% and the PSC with WBG perovskite absorber exhibits 0.97%, while their corresponding Quasi Fermi-level splitting (QFLS) values are calculated to be 1.159 eV for the PSC with NBG perovskite absorber and 1.289 eV for the PSC with WBG perovskite absorber, corroborate with the obtained VOC from the J–V scans. Furthermore, the PSC exhibits an integrated JSC of 25.7 mA cm−2 (Fig. 14h) with the NBG perovskite absorber with a calculated bandgap of 1.54 eV, the PSC exhibits a JSC of 22.1 mA cm−2 with the MBG perovskite absorber (bandgap: 1.59 eV), and the PSC exhibits a JSC of 18.3 mA cm−2 with the WBG perovskite absorber (bandgap: 1.77 eV).
a A schematic diagram illustrates the charge-carrier transport across the electron-selective layer/YbOx/Cu interface, where the carrier transport in the buffer layer (YbOx) has been thermally activated, hopping to another state from one localized state. b Schematic diagram depicts the device configuration of a p-i-n PSC with the buffer layer (YbOx), where the magnified area shows the amorphous nature of the buffer layer (ESL Electron-selective layer, HSL Hole-selective layer, TCO Transparent conducting oxide). The J–V characteristics of the p-i-n PSCs with c NBG perovskite, d MBG perovskite, and e WBG perovskite (Inset: SPO of the devices under simulated AM 1.5 G illumination at the maximum power point); f Distribution of PCE for PSCs with various bandgaps perovskites (NBG perovskite, MBG perovskite and WBG perovskite), where 15 devices are used at each bandgap; g The VOC of p-i-n PSCs with varied bandgaps perovskites using YbOx buffer layer in comparison with the other reported p-i-n PSCs with the BCP buffer layer (Dashed line: 90% of the radiative limit concerning the energy of the bandgap); h The EQE spectrum of the p-i-n PSC with NBG perovskite in comparison with MBG and WBG perovskites (reproduced with permission from ref. 73 Copyright 2024, Springer Nature).
Utilization of robust heterointerfaces for carrier viaducts through interfacial molecular bridges can enhance charge carrier extraction and transport, strengthen weak interface, preserve contact stack, act as a double-sided molecular binder, and that can enhance the performance of the PSCs. Xu et al.184 have fabricated robust heterointerfaces for carrier viaducts through interfacial molecular bridges to enable charge carrier selectivity via suitable energy level alignment, electrostatics, and low interfacial recombination to enhance the performance of PSCs, where the interfacial molecular bridge is composed of Ph-CH2N+H3−n(CH3)n ammonium cations (n: degree of substitution). The experimental and theoretical studies disclose that the heterointerface with the highest stability is achieved through quaternary ammonium (n = 3), where the phenyl groups are oriented vertically downward toward the perovskite substrates, and the -N+(CH3)3 groups selectively integrate into the perovskite frameworks. By acting as a carrier viaduct, this interfacial molecular bridge structure redistributes an uniform atmosphere at the heterointerface and allows for directional carrier management. Consequently, in both in-plane and out-of-plane directions, charge carrier extraction and transport are improved by the carrier viaduct approach. In the meantime, the weak interface is strengthened and the contact stack is preserved by the bottom interfacial molecule, which functions as a double-sided molecular binder. As a result, the PSC with the 1.54 eV bandgap perovskite absorber exhibits a high PCE of 25.45% with a fill factor of 85.66% (95% of the thermodynamic limit at its bandgap), and the perovskite solar module exhibits a high PCE of 20.91% (23.25 cm2), while the unencapsulated PSC exhibits high stability (>92% retention for 3000 h under light soaking by MPP tracking test).
Introduction of the suitable macrocyclic molecule as an interlayer between ETL and perovskite can enhance the charge separation and charge transfer, afford favorable energy level alignment and diminish charged defects, and that can enhance the performance of the PSCs. He et al.185 have utilized the macrocyclic molecule valinomycin as an interlayer between ETL and perovskite to alleviate both positively and negatively charged defects and enhance the performance of PSCs. The experimental and theoretical studies disclose that valinomycin is composed of multiple active sites of ─NH, ─C═O, and ─O─, where the positively charged defect can be managed by immobilizing the uncoordinated Pb2+ with the ─O─ and ─C═O groups in valinomycin, while the negatively charged defect associated with the formamidine vacancies can be obviated by the generation of N─H···I hydrogen bonding. The valinomycin interlayer exhibits favorable downshift band bending at the interface of perovskite and ETL, and thus charge separation and charge transfer are enhanced. Thus, the PSC with valinomycin affords favorable energy level alignment and diminishes charged defects. Consequently, the PSC with valinomycin exhibits a high PCE of 24.06%, while it exhibits high stability.
Utilizing interface and bulk engineering strategy can tune the bottom charge transporting layer/perovskite interface, which can improve the radiation of the full device stack, and that can enhance the performance of the PSCs. Jeong et al.186 have enhanced the radiation of the full device stack by tuning the bottom CTL/perovskite interface (CTL: charge transporting layer) through the interface and bulk engineering to enhance the radiation limit of PCE, while the correlation of the PCE of the device with the external PLQE of the full device stack is provided (PLQE: photoluminescence quantum efficiency). As a result, the device exhibits a high external PLQE of 15.57%, the PSC exhibits a high PCE of 26.04%, and it exhibits about 95% Shockley-Queisser limited Voc while it exhibits high stability.
Utilization of buried-metal-grid tin-doped indium oxide can decrease the power loss caused by the unfavorable high sheet resistance of the transparent conducting oxide electrode, which can enhance the performance of parallel-connected PSCs. Li et al.187 have designed the BMG ITO electrode to diminish the power loss caused by the unfavorable high sheet resistance of the transparent conducting oxide electrode to enhance the performance of parallel-connected PSC (BMG ITO: buried-metal-grid tin-doped indium oxide). The BMG ITO electrode is prepared through a photolithography technique by burying a metal grid (thickness: 140 nm) into ITO, where the sheet resistance of the BMG ITO electrode is 2.7 Ω sq−1, which is much less than that of 15.0 Ω sq−1 for ITO. Thus, the charge carrier transport of PSC is obviously tuned by the metal step of BMG over ITO, while the PSC with BMG ITO using a low metal step exhibits much higher PCE when compared to that of PSCs with BMG ITO with higher metal steps. Additionally, upon increasing the size of the active area of devices, the PSCs with BMG ITO exhibit a lesser decrease in PCE when compared to those with ITO. Hence, the parallel-connected PSCs exhibit excellent PCE of 24.3% and 22.5% (102.8 mm2), while the PSC exhibits high stability. This BMG ITO electrode strategy is also compatible for the construction of organic solar cells and inverted PSCs.
Strategies for interlayer/molecular bridge/buried-metal-grid electrodes, including the utilization of amorphous ytterbium oxide as a multifunctional and chemically stable buffer material between ETL and metal electrode73, the utilization of the macrocyclic molecule valinomycin as an interlayer between ETL and perovskite185, the utilization of buried-metal-grid tin-doped indium oxide187, modulation of the bottom CTL/perovskite interface to improve the radiation of the full device stack186, and fabrication of robust heterointerfaces for carrier viaducts through interfacial molecular bridges184 enhanced the efficiency and/or stability of the PSCs.
link